I recently revealed my shameful secret – that I don’t own a DSM-IV Manual and am only vaguely aware of what’s in it. Now I’ll extend my unsolicited self revelation. Although I’d heard the term and had some notion of its meaning, until yesterday I didn’t know what SCID [Structured Clinical Interview for DSM Disorders] stood for. And more, I didn’t know that people had actually applied the computer programming concept, Algorithm, to Psychiatric treatment. Nancy’s comment on the last post referred me to a couple of papers about an algorithmic computer program for the treatment of Major Depressive Disorder. The first paper [A Computerized Clinical Decision Support System as a Means of Implementing Depression Guidelines] described the program itself. The second paper [Barriers to implementation of a computerized decision support system for depression: an observational report on lessons learned in "real world" clinical settings] was a later [quaisi-Freudian] interpretation of why clinicians wouldn’t use it [unless someone was looking over their shoulders] – an interpretation of the resistance [assuming that not using the program is a sign of epidemic mental illness among clinicians].
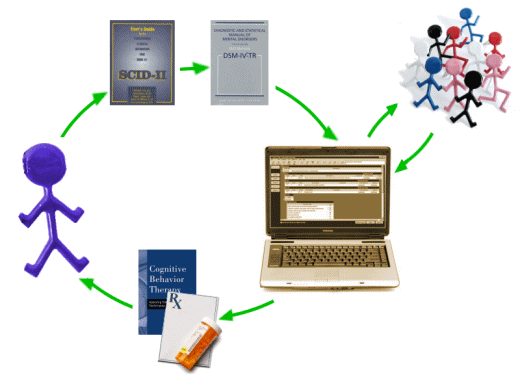
The short version goes like this: The patient [purple guy] is interviewed using the SCID [Structured Clinical Interview for DSM Disorders] which is a reproducible set of questions leading to a full DSM IV diagnosis in all Axes – then serving as the input for the CompTMAP [Computer – Texas Medication Algorithm Project] program. Based on the parameters entered, the program makes treatment recommendations from among the available modalities, including "tweaking" on follow-up visits. The algorithms are presumably derived from the sea of research and clinical trials referred to in my recent posts [particularly inspiring my whimsical NIMH·receptalator].
All of the elements in this system taken together are referred to as
EVIDENCE-BASED MEDICINE and, in some virtual future, could all be done with a computer or questionnaires. While one might question this assumption, each element is backed by statistically validated EVIDENCE – ergo
EVIDENCE-BASED MEDICINE. As mentioned, apparently they’re having trouble getting doctors to use
CompTMAP, and so the second paper delves into the possible reasons that this implementation of
EVIDENCE BASED-MEDICINE is not replacing
whatever-you-might-call-the-alternative. Here’s the problem in the words of
CompTMAP‘s
Dr. Madhukar H Trivedi:
Although state of the art evidence-based clinical guidelines have been available to clinicians for some time, it is now well understood that the implementation or adoption of research information in actual clinical practices lags far behind, with limited potential to change physician behavior. Unfortunately to date, efforts to implement quality improvement (QI) research findings into practice have often proceeded without insistence on the same level of rigor required to establish these QI targets as worthy of implementation. The capacity to base treatment decisions on state-of-the-art knowledge alone appears to be insufficient to motivate behavior change among clinicians. Despite this, passive dissemination of evidence-based approaches in the form of instructional workshops or written manuals is the most frequently used approach to implementation of evidence-based guidelines in real-world settings. While there is evidence that studies have demonstrated improved outcomes when guidelines are introduced during a research project, unfortunately benefits dissipate after the research program is over.
Why not? Why would perfectly reasonable Texas Psychiatrists stop using this tool as soon as Dr. Trivedi turns his back? Are they opposed to QI [Quality Improvement]? Well Dr. Trivedi has given the clinician’s behavior some serious thought:
A recent systematic review of the literature identified barriers to guideline adherence, including a lack of: awareness, agreement, or perceived self-efficacy to change; minimal outcome expectancy; and an inertia associated with faith in existing treatment practices . In addition, external barriers such as lack of time, insufficient staff/support, and patient-related factors were also listed. The authors concluded that it is important to be aware of these barriers so that they may be overcome. One essential factor in successful implementation is physicians’ faith in the algorithm itself, particularly in light of the common physician concern about losing autonomy in treatment decision-making.
Quite a list of possibilities, including
faith, not something one thinks about much in science. And then there’s the wounded ego factor ["
the common physician concern about losing autonomy in treatment decision-making"]. They go on to describe their various ideas about overcoming these barriers. Their discussion is in the
article for you to read yourself [it reminds me of my mother’s attempts to interest me in Broccoli and Cauliflower – vegetables I still consider to be members of the Tree Family].
I fully intend to devote several posts to offering alternative reasons that those Texas Psychiatrists might be resisting this state-of-the-art evidence-based tool that are more fundamental than those suggested in the referenced paper, but for the moment, I’d like to briefly address the last two that Dr. Trivedi mentions: the "physicians’ faith in the algorithm itself" and "the common physician concern about losing autonomy in treatment decision-making." Perhaps Dr. Trivedi might consider that this piece at the end of his paper has something to do with the problem:
Competing interests
Madhukar H. Trivedi, M.D. has been a consultant for Abbott Laboratories, Inc.; Akzo (Organon Pharmaceuticals Inc.); AstraZeneca; Bayer; Bristol-Myers Squibb Company; Cephalon, Inc.; Cyberonics, Inc.; Eli Lilly & Company; Fabre-Kramer Pharmaceuticals, Inc. Forest Pharmaceuticals; GlaxoSmithKline; Janssen Pharmaceutica Products, LP; Johnson & Johnson PRD; Eli Lilly & Company; Meade Johnson; Neuronetics; Parke-Davis Pharmaceuticals, Inc.; Pfizer, Inc.; Pharmacia &; Upjohn; Sepracor; Solvay Pharmaceuticals, Inc.; VantagePoint; and Wyeth-Ayerst Laboratories.
He has served on speakers bureaus for Abdi Brahim; Akzo (Organon Pharmaceuticals Inc.); Bristol-Myers Squibb Company; Cephalon, Inc.; Cyberonics, Inc.; Forest Pharmaceuticals; GlaxoSmithKline; Janssen Pharmaceutica Products, LP; Eli Lilly & Company; Pharmacia &; Upjohn; Solvay Pharmaceuticals, Inc.; and Wyeth-Ayerst Laboratories.
He has also received grant support from Bristol-Myers Squibb Company; Cephalon, Inc.; Corcept Therapeutics, Inc.; Cyberonics, Inc.; Eli Lilly & Company; Forest Pharmaceuticals; GlaxoSmithKline; Janssen Pharmaceutica; Merck; National Institute of Mental Health; National Alliance for Research in Schizophrenia and Depression; Novartis; Pfizer Inc.; Pharmacia &; Upjohn; Predix Pharmaceuticals; Solvay Pharmaceuticals, Inc.; and Wyeth-Ayerst Laboratories.

“The capacity to base treatment decisions on state-of-the-art knowledge alone appears to be insufficient to motivate behavior change among clinicians.”
It’s amazing how scientific statements, when taken out of context (or, in this case, even IN context) can be rather insulting.
Q.E.D.!
Ouch! I didn’t see that coming.
“The capacity to base treatment decisions on state-of-the-art knowledge alone appears to be insufficient to motivate behavior change among clinicians.â€
Seems a sort of re-working of the old saw about leading a horse to water (and a tad patronizing). Sometimes the horse won’t drink because he knows darned well that there is something dangerous to his homeostasis in the water, sometimes he isn’t really thirsty, never would he develop a kind of neurotic petulance to refuse the “evidence” that water was available to him. Horses (and I’d hazard, most clinicians) know better.
I’ve not yet read the paper but it seems Dr. Trivedi has simply not made a sufficient case…just because he’s built it doesn’t mean every clinician and their kid brother will be beating a path to his door ( further complicated by his regular drinking from the corporate kool aid fountain).
On the face of it, these complicated diagnostic systems have been touted for some time – they have origins in “AI” and the extraordinary increases in data processing power since the mid-80s anyway. The keyword, in my humble opinion has always been “Artificial”.
Carl,
What a great thought. It is A.I., and it is very “artificial”. I’m stealing that line!…