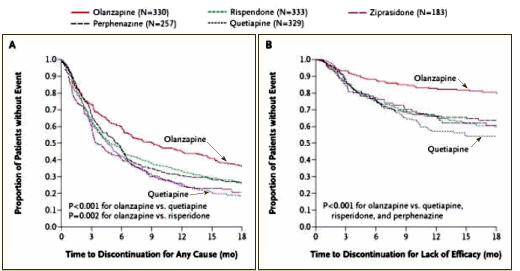
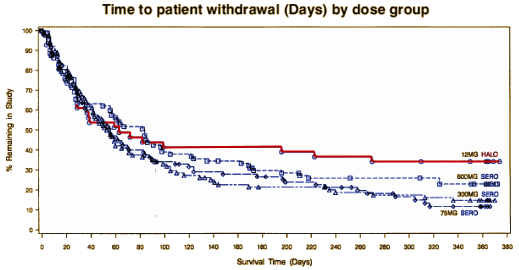
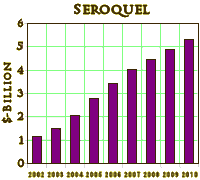
 In an earlier series, I mused guiltily that had I been on the F.D.A. Panel evaluating Seroquel, I might’ve thought, "No great shakes, but let’s give ’em a shot. If it’s no good, doctors won’t prescribe it and the patients won’t take it." Well that didn’t happen. CATIE showed what we would’ve known from Trial 0015 [had we seen it], the Schizophrenic patients stopped taking it over the other choices, new drugs and old. But the sales of Seroquel had risen and would continue to rise. While it’s tempting to look into the issue of weight gain, for the moment I’m going to stick with the sales side of the equation. It’s obvious that this drug isn’t a personal favorite, but I hope I laid out the basics fairly. The graph on the right is not explained by Seroquel‘s stellar performance in Schizophrenia. In fact, a widely known study like CATIE didn’t slow the sales of Seroquel at all, because by 2005, their market share likely didn’t have much to do with it being used to treat Schizophrenia.
In an earlier series, I mused guiltily that had I been on the F.D.A. Panel evaluating Seroquel, I might’ve thought, "No great shakes, but let’s give ’em a shot. If it’s no good, doctors won’t prescribe it and the patients won’t take it." Well that didn’t happen. CATIE showed what we would’ve known from Trial 0015 [had we seen it], the Schizophrenic patients stopped taking it over the other choices, new drugs and old. But the sales of Seroquel had risen and would continue to rise. While it’s tempting to look into the issue of weight gain, for the moment I’m going to stick with the sales side of the equation. It’s obvious that this drug isn’t a personal favorite, but I hope I laid out the basics fairly. The graph on the right is not explained by Seroquel‘s stellar performance in Schizophrenia. In fact, a widely known study like CATIE didn’t slow the sales of Seroquel at all, because by 2005, their market share likely didn’t have much to do with it being used to treat Schizophrenia.

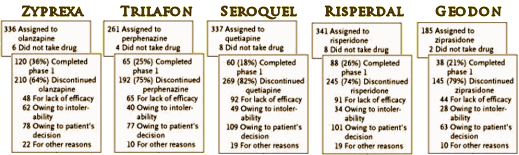
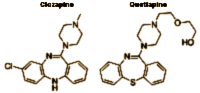 Zeneca had poured years and a lot of money into getting their drug approved. Now it was time to focus on reaping the benefits of their hard work. This email mentions Seroquel‘s starting place – Clozaril [Clozapine]. Clozapine was the first Atypical Antipsychotic. It had been around for decades. It is a potent antipsychotic that has less neurotoxicity [EPS or Tardive Dyskinesia] than the first generation neuroleptics, but has other big problems – weight gain, Diabetes, and Bone Marrow toxicity [Agranulocytosis] which can be fatal. Schizophrenia can be a malignant disease. The early name was Dementia Praecox, and one of the characteristics was an early death. In some patients, Clozapine works when all others fail, and in an intensely suicidal or catatonic patient, the risks are offset by the potential gain. So the drug is available with strict monitoring of the Bone Marrow status – rarely used. But Clozapine has remained the marker – a drug with less neurotoxicity and potent antipsychotic prowess. Thus, it has been the force propelling the development of the Atypical Antipsychotics – searching for a "safe Clozapine." The fact that there is a more potent drug is a beacon for researchers to find one that can be used safely, and routinely.
Zeneca had poured years and a lot of money into getting their drug approved. Now it was time to focus on reaping the benefits of their hard work. This email mentions Seroquel‘s starting place – Clozaril [Clozapine]. Clozapine was the first Atypical Antipsychotic. It had been around for decades. It is a potent antipsychotic that has less neurotoxicity [EPS or Tardive Dyskinesia] than the first generation neuroleptics, but has other big problems – weight gain, Diabetes, and Bone Marrow toxicity [Agranulocytosis] which can be fatal. Schizophrenia can be a malignant disease. The early name was Dementia Praecox, and one of the characteristics was an early death. In some patients, Clozapine works when all others fail, and in an intensely suicidal or catatonic patient, the risks are offset by the potential gain. So the drug is available with strict monitoring of the Bone Marrow status – rarely used. But Clozapine has remained the marker – a drug with less neurotoxicity and potent antipsychotic prowess. Thus, it has been the force propelling the development of the Atypical Antipsychotics – searching for a "safe Clozapine." The fact that there is a more potent drug is a beacon for researchers to find one that can be used safely, and routinely.
AstraZeneca Said to Pay $150 Million to Settle Seroquel Suits
Bloomberg Business Week
By Jef Feeley
February 17, 2011AstraZeneca Plc agreed to pay $150 million to settle more lawsuits claiming its antipsychotic drug Seroquel causes diabetes, pushing the amount the drugmaker has paid to resolve cases over the medicine to almost $350 million, people familiar with the accords said. AstraZeneca, the U.K.’s second-biggest drugmaker, will resolve about 6,000 cases alleging the company knew Seroquel could cause diabetes and failed to adequately warn patients, two people familiar with the settlements said. They spoke on the condition of anonymity because they weren’t authorized to speak publicly about the accords. The cases settled for an average of about $25,000, the people said.
The settlements signal AstraZeneca is seeking to put the Seroquel litigation behind it as it works to overcome setbacks in its drug-development pipeline, said Jeremy Batstone-Carr, London-based analyst for Charles Stanley & Co., who rates the drugmaker’s shares as “accumulate.” “Legal cases represent one of the great imponderables that can act on shareholder sentiment,” Batstone-Carr said. “You try to clear the decks and get investors as great a degree of certainty as possible.”
The settlement, which resulted from a court-ordered mediation, leaves AstraZeneca now facing only about 4,000 Seroquel claims, the people said. The London-based drugmaker announced last summer it had resolved about two-thirds of the 26,000 suits over the drug that had been filed in courts around the U.S…
I think you’ll enjoy this satire from pharmagiles
http://pharmagiles.blogspot.com/2011/02/tiny-minded.html
And understand when any AZN/pharma stock analyst such as Jeremy Batstone-Carr of London-based Charles Stanley & Co. makes any comment regarding his specific investment sector of which we already know is a handful of the largest drug companies, he too is bound to report as positive a report possible since that is what he is paid to do.
They have to find something positive to say of the average investor hanging on these crooks every word will kill the stock price if I have to remind anyone stocks and the companies they are issued from once in the hands of investors and speculators trade by perception alone as you see here it is always forward looking.
proof of what Im saying is no analyst ever comes out with a sell or sell short rating which in 2008 would have saved many innocent people’s retirement funds, but instead what he is doing here is releasing an “accumulation” statement.
I interpret that to mean short term he sees the stock going down so some geniuses out there with extra pocket change will be buying it up as it drops. they will rationalize this pending huge dividend remains intact. when in fact the big dividend is just a bribe to keep investors holding the stock.
As the stock price goes lower it will become an attractive buy because then the return goes up from the dividend. anyway its all a scam how this thing works, but I take his statement as a veiled signal to a few in the know where he thinks its going. It wont get crushed but its going down on some really bad new AZN is scrambling around trying to quash.