The CROs [Clinical Research Organization AKA Contract Research Organization] aren’t visible. They’re a mixture of Consultant/Contract Units so they don’t show unless you know how to look for them. For example, Parexel has been all over these posts I’ve been writing about Seroquel, but I didn’t know it when I was writing them [because I didn’t know what a CRO was]. This post goes back over those posts for a look at the CRO‘s footprints:
Clinical Research Trials
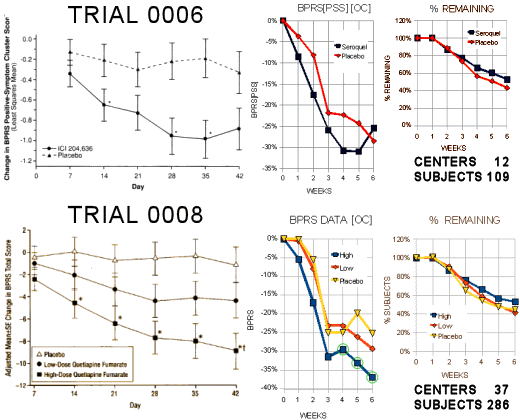
The above is from their first two submitted Seroquel Clinical Trials. As best I can tell, they were managed by Zeneca:
Accepted for publication November 6, 1996. This study was supported by a grant from Zeneca Pharmaceuticals, Macclesfield, England, and Wilmington, Del. We thank Suzanne Bristow-Marcalus for medical writing support; Jean Fennimore, RN, and Alison Smith, MSc, for trial organization and management support; Gary Cooper, MS, Jacqueline Fiore, Lesley Farrow, and Barney Home for trial monitoring support; and Kathleen Jelliffe, Denise Redkar-Brown, Joy Russo, and Thais Womack for data management support.
The writer is acknowledged in a number of
Zeneca articles and lives in Wilmington. The other people acknowledged that I can run down worked for
Zeneca. So I can’t get the real scent of a
CRO back in 1997. While
Trial 0006 was published with success claimed in the graph and write-up, looking at the uncorrected data [from the FDA document], it was not significant – actually, it was a bust. While the second shot [
Trial 0008] wasn’t that much more impressive, it did achieve significance and was the main workhorse that got the
Seroquel approved by the FDA [barely]. Notice that it uses more
CRCs [12 US -> 37 US and Europe] and it has a much larger cohort [109 -> 286]. That’s what it takes to get approved – statistical significance – and to get that with these small differences, you need the big numbers. By 2002,
Zeneca was
AstraZeneca and they hooked up with
Parexel, a large
CRO:
PAREXEL International Corporation, one of the world’s largest biopharmaceutical outsourcing companies, today announced that it has entered into a long-term preferred provider position for global clinical trials development and management services with AstraZeneca International, one of the world’s leading pharmaceutical companies. PAREXEL was awarded the status of Preferred Provider following an in-depth evaluation of its therapeutic expertise, worldwide operations, and knowledgeable experts. This alliance gives AstraZeneca access to PAREXEL’s comprehensive portfolio of biopharmaceutical services, spanning pre-clinical through regulatory approval as well as marketing, medical education, web-based technology solutions and process improvement…
This graph is from one of the
Seroquel in Bipolar Disorder studies [
TRIAL 0135] published in 2006 [BOLDER II ]:
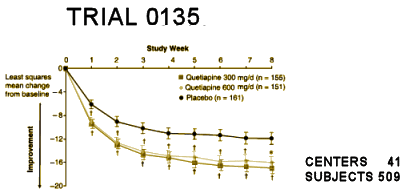
A different patient population and a different scale, but it has that familiar CRO-Chart look with a smooth drooping placebo curve and small differences. There are, however, some things to note. First, those little lines above and below the data points are the standard error of the mean, just like in the Schizophrenia trials [above] from 10 years before, but they’re much narrower. And those little symbols below the data points mark significant values – all data points achieve significance. Is Seroquel more effective in Bipolar Disorder than Schizophrenia? Of course not. This tighter data is because of the larger number of subjects [509]. If you want significance from small differences, you need a really big study [requiring more CRCs]. That’s one way you can tell a CRO is in the picture. Getting the subjects for the studies and producing that kind of significance is what CROs do. Remember, they’re in the "time-to-market" business.
Documents: Analysis and Writing
Here’s another clear footprint left by the CRO in that last trial:
The authors would like to acknowledge the editorial assistance of Aruna Seth, PhD [PAREXEL MMS]. Financial support for this assistance was provided by AstraZeneca Pharmaceuticals LP.
Parexel conducted this study [I expect that Aruna Seth of
Parexel wrote the article, but that part is not directly provable]. But remember that episode with
Dr. Nassir Ghaemi? when he was asked to be a second author on a paper? the one where he wanted to see the raw data and actually participate in writing the paper [
selling seroquel VII: indication sprawl…]? Well, the involved characters were from
Parexel.
Claudia Piano was in charge of the AstraZeneca Unit at
Parexel:
So at the time of the Ghaemi incident, Donna Casparro was in charge of Parexel‘s Seroquel Publications:
Notice that
Dr. Ghaemi is responding to
Parexel‘s
Donna Casparro, and
Dr. Roger McIntyre is forwarding his response to her, and she’s sending it to
AstraZeneca. By the way,
Parexel‘s
Aruna Seth is in the email loop and was acknowledged in
Dr. McIntyre‘s article [
selling seroquel VII: indication sprawl…]. What that means to me is that
Parexel was in charge of recruiting the authors for the
Parexel-generated papers from the
Parexel-run Clinical Trials [
the real ghosts in this story are the listed authors]. My assumption would be that the recruited "authors" at the most only reviewed
Parexel‘s galley proofs [Notice
McIntyre‘s comment, "… it is AZ data"].
Promoting the Brand
I guess that writing the articles and recruiting the authors is what they mean when the advertise Clinical Trial Development, but the CRO‘s services can go much further than just things related to Clinical Trials. Here’s a presentation proposal from Parexel to prepare four abstracts for the 2003 USPMHC to promote the "off-label" use of Seroquel in Bipolar Disorders [budget redacted] – before FDA approval:
And a similar Parexel-generated set of programs at the 2004 APA Meetings:
Here’s another to prepare a Parexel-generated abstract to promote Seroquel for treating Bipolar Disorders in Children and Adolescents [5 years before FDA approval for that indication]:
KOL [Key Opinion Leader]
MSL [Medical Science Liason]
In the
Seroquel unsealed documents, I can’t find much about how
Parexel is involved in
AstraZeneca‘s relationship with their bevy of
Key Opinion Leaders. My guess is that they have at least been involved in
KOL Communications and in preparing the materials that
KOLs use. Looking on the new
ProPublica Dollars-for-Docs site,
AstraZeneca pays about 2500 speaker-physicians in the vicinity of
$40 M/year [
I may be internally resistant to finding out about that, in part, because the KOL thing makes me so damned angry – but I did try]. By the way,
MSL is a new profession – people who locate and handle
KOLs. Here’s what
Parexel says on its web site:

This post was just meant to show by example what the CROs do – and one of those things is ghostwrite their Clinical Trials. I have no doubt about that. They also do promotion, and write the abstracts and posters used in that enterprise. I suspect that they coordinate and script the KOL activities, but I couldn’t find direct evidence for that activity in the Seroquel unsealed documents. But there are other things that are less tangible. Like this data from a previous post [selling seroquel VII: indication sprawl…]:
| CLINICAL TRIALS |
| |
2001 |
2002 |
2003 |
2004 |
2005 |
2006 |
2007 |
2008 |
2009 |
2010 |
| |
|
| Schizophrenia |
1 |
|
2 |
5 |
2 |
|
2 |
2 |
1 |
2 |
| Bipolar Disorders |
|
3 |
|
6 |
4 |
2 |
2 |
|
3 |
2 |
| Major Depression |
|
|
|
|
2 |
8 |
1 |
2 |
1 |
|
| Anxiety Disorder |
|
|
|
1 |
|
5 |
1 |
1 |
2 |
|
| Other |
|
1 |
1 |
4 |
4 |
|
2 |
3 |
3 |
|
Parexel signed on with AstraZeneca in 2002. Looking at the Table of Clinical Trials, it looks as if they got right to work [generating more Clinical Trials business for themselves and indication approvals for AstraZeneca]. That in turn generated launch campaigns and brand promotions for Parexel – quite a marriage. They had some rough going when it turned out that one of AstraZeneca‘s big-mojo Docs [Wayne MacFadden] was having an affair with one of Parexel‘s big mojo writers, and was pumping her for info about a competitor who was also doing business with Parexel. Such things.
I don’t know if Parexel is still doing AstraZeneca‘s Clinical Trials, but somebody is [for Seroquel XR], and this is the most recent one in clinicaltrials.gov – something we’ll talk about later – "globalization."
| Evaluate the Efficacy and Safety of Quetiapine Fumarate (SEROQUEL) Extended Release as Monotherapy in the Treatment of Patients With Bipolar Depression |
Please refer to this study by its ClinicalTrials.gov identifier: NCT01256177
Contact: AstraZeneca Clinical Study Information – Huafei.Li@astrazeneca.com
|
| China, Beijing |
recruiting |
Beijing, Beijing, China |
| China, Guangzhou |
recruiting |
Guangzhou, Guangzhou, China |
| China, Hebei |
recruiting |
Baoding, Hebei, China |
| China, Hubei |
recruiting |
Wuhan, Hubei, China |
| China, HuNan |
recruiting |
Changsha, HuNan, China |
| China, Jiangsu |
recruiting |
Nanjing, Jiangsu, China |
| China, Shanghai |
recruiting |
Shanghai, Shanghai, China |
| China, Shanxi |
Not yet recruiting |
Taiyuan, Shanxi, China |
| China, Sichuan |
Not yet recruiting |
Chengdou, Sichuan, China |
| China, Tianjin |
recruiting |
Tianjin, Tianjin, China |
| China, Yunnan |
recruiting |
Kunming, Yunnan, China |
| China, Zhejiang |
Not yet recruiting |
Hangzhou, Zhejiang, China |
I apologize for the disjointedness of this post. I wanted to go through this specific example where we have the most inside information to see what the CROs actually do in practice. I expect I missed many clues, but I found enough to let me know that they do a lot. They keep a low profile. There’s apparently no requirement that their presence be specifically acknowledged except as "writing assistance."
What I personally think is that the CROs are the ghosts in the machine, the unseen intelligence that speeds a drug to market and then maximizes its profitability throughout its patent life – all in the background. As I’ve been researching this particular piece of the story, I have to stop periodically. More than the other of the catch-up series I’ve been doing, this one makes me the maddest. I’m not yet sure of all the reasons – but part of it is the secretiveness. It seems to me that their entire reason for being is unseen corruption – corruption of science, of medicine, of doctors, of patients, and even of their pharmaceutical clients. I need to think about that more, but before looking at the CRO industry as a whole, I might take a break and read an uplifting novel, or maybe a biography of Mother Teresa to clean out my soul.









..”AstraZeneca and they hooked up with Parexel.”
well, this post is positively loaded w info.
Along w martha rosenberg (columnist) i was threatened w a major lawsuit for inferring MacFadden had an affair w a certain woman for divulging trade secrets via sex favors. had to pull down the post and place a mea culpa (their words) on the blog for 30 days.
I found the woman’s name in rosenberg’s article and linked to it.
The more you get into this topic the more you will feel the need to take a long walk or watch cartoons to clear the mind.
Great work, I can barely keep up with reading!
http://www.fiercebiotech.com/story/chinas-cro-industry-booms-pharma-demand-heats/2009-09-03?sms_ss=twitter&at_xt=4d746bc79cfbd035,0
FYI
Cheers
Remember AZ outsourced main ingredient lactam in Seroquel=made in China!
http://bipolarsoupkitchen-stephany.blogspot.com/2010/10/made-in-china-astrazeneca-buys-lactam.html
btw Seroquel XR trials? are at University of MN conducted by Dr Charles Schulz sponsored by AstraZeneca –same place CAFE trial and Dan Markingson suicide story is based…the XR is being trialed for Borderline Personality Disorder.