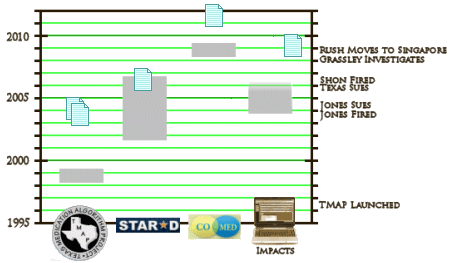
Who ever knows how history will be written in the future when it longer holds us in its grip. Will the Age of Corruption start with TMAP, maybe George W. Bush, Reagan’s Deregulation, Nixon’s Watergate, or maybe go back to LBJ’s decision to fabricating the Gulf of Tonkin? I doubt that TMAP and its spawn will deserve even a paragraph in this chapter [though the pharmaceutical invasion of psychiatry after 1980 may be a contender for honorable mention]. But from right here, it’s still a pretty big story what happened in Texas in 1996 with the Texas Medical Algorithm Project.
 Dr. Rush arrived in Dallas around 1978 as a Cognitive Behavior expert. By the time Dr. Trivedi showed up [Trivedi’s first paper] in 1993, they were collaborating on a biology of depression paper. In 1996, they were both involved in setting up TMAP [Texas Medical Algorithm Project] for the State of Texas – a joint project of UT Southwestern, Pharmaceutical contributions, and the Texas Mental Health system. On the face of things, it seemed benign. The primary task was to create a Board to decide what medications would be approved for the vast public system in Texas [Medicare, Medicaid, Prisons, Mental Health Centers]. The twist was the idea to have treatment guidelines, or algorithms, to standardize and maybe optimize practice. Sounds reasonable. The program came at a time when there weren’t any options for generic SSRIs, but it was on the leading edge of the wave of atypical antipsychotics at a time when hopes were high for those drugs [as were prices] to replace the older neuroleptics. So it was no surprise that the atypicals appeared on the Texas State formulary. From Allen Jones:
Dr. Rush arrived in Dallas around 1978 as a Cognitive Behavior expert. By the time Dr. Trivedi showed up [Trivedi’s first paper] in 1993, they were collaborating on a biology of depression paper. In 1996, they were both involved in setting up TMAP [Texas Medical Algorithm Project] for the State of Texas – a joint project of UT Southwestern, Pharmaceutical contributions, and the Texas Mental Health system. On the face of things, it seemed benign. The primary task was to create a Board to decide what medications would be approved for the vast public system in Texas [Medicare, Medicaid, Prisons, Mental Health Centers]. The twist was the idea to have treatment guidelines, or algorithms, to standardize and maybe optimize practice. Sounds reasonable. The program came at a time when there weren’t any options for generic SSRIs, but it was on the leading edge of the wave of atypical antipsychotics at a time when hopes were high for those drugs [as were prices] to replace the older neuroleptics. So it was no surprise that the atypicals appeared on the Texas State formulary. From Allen Jones:
TMAP concluded that all newer, patented anti-depressants were superior to generics.
TMAP concluded that the patented bi-polar drugs were superior to generic drugs.
TMAP concluded that “Expert Consensus” established these drugs to be safer, more effective, better tolerated and relatively free of side effects when compared to the older, generic, medications.
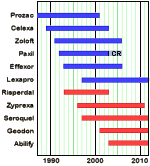
TMAP then formulated separate “algorithms” (flow charts) and drug menus for the treatment of schizophrenia, depression and bi-polar disorder. All of the new, patented drugs were incorporated into the TMAP algorithms.
State doctors following the algorithms were and are required to use these drugs. The administrative decision of a State Mental Health Program to adopt TMAP brought with it the clinical decision to use the recommended drugs on all patients in the state system. A state doctor may choose which patented drug to use, but he may not choose to use a generic drug until at least two, often three, patented drugs have failed.
John Rush directed TMAP assisted by psychopharmacologist Steven Shon. Madhukar Trivedi headed the Depression section. They set about researching their treatment algorithm for Major Depressive Disorder first thing in 1998. Their study was something of a John Henry versus the machine affair – pitting public clinics using their complex algorithms [ALGO] against treatment as usual clinics [TAU]. This time, the machine [ALGO] won [though the results were disappointing overall]. They quickly designed a $35 M study for a similar enterprise through the NIMH using a modified algorithm. They published a sea of articles on TMAP, but the two definitive articles didn’t come out until after STAR*D [the NIMH study] was underway. One article [Trivedi] was about John JHenry’s defeat. The second explained the low response rates as being due to their chronic impoverished cohort of patients. STAR*D was aimed at a higher class clientele. Steven Shon was also on the move, jetting around the country and the world pitching TMAP [as it turned out, on the pharmaceutical dollar]. He engaged 16 states in adopting some version of the program. Drs. Rush and Trivedi were being well reimbursed for their active participation in pharmaceutical paid speakers bureaus, scientific advisory boards, and continuing education presentations. Meanwhile, the Texas public medications budget was nearing bankruptcy.
All told, these projects which produced no usable information cost the NIMH around $50 M, and the State of Texas much more [Billions?]. Trivedi’s IMPACTS [CDDSS-D] never made it off the ground. TMAP is long abandoned. CO-MED was a bust all around. And STAR*D would best be retracted as simply bad science with an unreadable outcome. As for John Rush, he’s in Singapore doing whatever he does there, but is involved in Brain Resources personalized medicine project. Trivedi is Principle Investigator in an NIMH study of the same thing – personalized medicine. It’s a new day with a new fad.
It is no mystery to me. Charles Nemeroff and Tom Insell. Enough said.
I’ve fallen way behind in this general discussion but I thought we solved Trivedi et al months ago! Isn’t he the twit who was in a huff because medical doctors didn’t behave like automatons in the “real” world?