Paxil ad from 2001
Paxil ad from 2002
I just received a copy of a GSK Press Release from 2002, obviously published in response to the ruling in the last post. It speaks to a lot of things, but the central point is how corrupt things had become by then. They still had four years of patent protection ahead of them, and they were determined to milk it for all it was worth. Notice the language highlighted below, also the Key Opinion Leaders available for comment:
|
Philadelphia, PA, August 20, 2002
|
|
GlaxoSmithKline (NYSE: GSK) today announced it will appeal a ruling by a U.S. District Court judge in Los Angeles requiring the company to alter its television advertising for the anti-depressant Paxil® (paroxetine hydrochloride) that states that Paxil is not habit forming.
"We are very disappointed in the ruling. The US Food and Drug Administration (FDA) – and not the courts – has the expertise and responsibility for reviewing and regulating pharmaceutical ads," said David Stout, president of U.S. Pharmaceuticals at GlaxoSmithKline. "The Paxil television ad was submitted for FDA review prior to use, and the Agency raised no objections to the language at issue." "GSK strongly stands behind the safety and efficacy of Paxil," said Stout. "Physician organizations like the American Psychiatric Association have stated that antidepressants are not habit forming. It is also important to note that the Court has made no finding that Paxil is addictive or induces dependency." Paxil is a leading selective serotonin reuptake inhibitor (SSRI) indicated for the treatment of depression and all major anxiety disorders including social anxiety disorder, panic disorder, obsessive compulsive disorder, generalized anxiety disorder and post-traumatic stress disorder. GlaxoSmithKline, one of the world’s leading research-based pharmaceutical and health care companies, is committed to fighting disease by bringing innovative medicines and services to patients and to the health care providers who serve them. U.S. headquarters are in Philadelphia, PA, and Research Triangle Park, N.C.
|
| Editor’s Note: |
|
The following physicians are available for comment. To arrange an interview, please contact Sherri Jaffe at 914 656 5479.
Charles B. Nemeroff MD, PhD, Reunette W. Harris Professor and Chairman, Department of Psychiatry and Behavioral Sciences, Emory University School of Medicine Alan F. Schatzberg MD, Professor and Chairman, Department of Psychiatry and Behavioral Sciences, Stanford University Medical School Vivien K. Burt MD, PhD, Professor of Clinical Psychiatry, UCLA School of Medicine; Director, Women’s Life Center, UCLA Neuropsychiatric Institute and Hospital David V Sheehan MD, MBA, Professor of Psychiatry, University of South Florida College of Medicine, Tampa Florida "Let’s Talk Facts About Depression," American Psychiatric Association, http://www.psych.org/public_info/depression.cfm
|
In the 2001 ad above, they say, "Paxil is non-habit forming." In the 2002 version, they say, "Paxil is non-habit forming," but add, "Don’t stop taking Paxil before talking to your doctor." Those are the ads that lead to the ruling referred to in the press release:
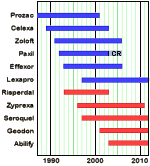 The person that got me focused on this topic actually started on Paxil in 2002 when she was 14 years old. By 2002, withdrawal obviously wasn’t well enough known publicly to keep people from starting patients on it. In 2002, the Internet wasn’t full of personal reports, e-books about getting off of it, or youtube advice videos. At that time, Paxil was in its prime. Drs. Nemeroff and Schatzberg had signed on to a ghost-written textbook a couple of years before that was being handed out to Primary Care Physicians leaning doctors towards Paxil. Dr. Martin Keller et al had just published a paper claiming it was effective in depressed adolescents. That was the infamous Study 329 now widely discredited that failed to mention the akisthisia and the suicidal thoughts in that age group. 2002 was also the year when the BBC program, Panorama, did a series on Paxil [Seroxat] in October. The program received 1400 emails in response [still available on their website] and was a wake-up call for many. ABC Primetime did a similar series here in 2004 [ Drug Maker Withheld Paxil Study Data ]. But 2002 was still an arrogant time at GSK, in spite of the growing disquiet.
The person that got me focused on this topic actually started on Paxil in 2002 when she was 14 years old. By 2002, withdrawal obviously wasn’t well enough known publicly to keep people from starting patients on it. In 2002, the Internet wasn’t full of personal reports, e-books about getting off of it, or youtube advice videos. At that time, Paxil was in its prime. Drs. Nemeroff and Schatzberg had signed on to a ghost-written textbook a couple of years before that was being handed out to Primary Care Physicians leaning doctors towards Paxil. Dr. Martin Keller et al had just published a paper claiming it was effective in depressed adolescents. That was the infamous Study 329 now widely discredited that failed to mention the akisthisia and the suicidal thoughts in that age group. 2002 was also the year when the BBC program, Panorama, did a series on Paxil [Seroxat] in October. The program received 1400 emails in response [still available on their website] and was a wake-up call for many. ABC Primetime did a similar series here in 2004 [ Drug Maker Withheld Paxil Study Data ]. But 2002 was still an arrogant time at GSK, in spite of the growing disquiet.
I first took Seroxat [Paxil in the USA] in 1997. I finally decided to stop taking it in May 2004 and it then took me 22 months to slowly wean off the drug. All in all I have lost almost 10 years of my life to this drug. I want to share with you what I’ve found out since May 2004. I’ve discovered how dangerous SSRIs can be and how dangerous the big drug companies are – especially GlaxoSmithKline [who make Seroxat].
I think at this point I must mention the drug regulators here in the UK [The MHRA] and the FDA in America. If you’re new to all this you might think they are the organisations that we can trust to keep us safe from nightmare drugs like Seroxat. Not the case I’m afraid.
The public has been let down by them on all too many occasions. I’ve become very cynical since May 2004 and as I’ve learnt more and more and I have to say it looks from here as if the MHRA and The FDA are more concerned with protecting the Pharmaceutical companies than protecting the public. There’s a lot of money to be made from selling new drugs to the world – a lot of money indeed. It doesn’t seem to matter if the drugs don’t quite work as well as they should or if people can become addicted to them. In the case of Seroxat, Glaxo has simply hidden trial data that shows how dangerous Seroxat really is. In this Blog I’ll be naming and shaming the guilty and I’ll be telling the truth about Seroxat.
Here’s what Dr. David Healy had to say about Paxil Withdrawal in his 2004 book, Let Them Eat Prozac [page 270]:
YES keep bringing it up, because doctors still hate to talk abt psych meds being addictive or have withdrawals, inc how once OFF the SSRI, it can BE WORSE for a few months or longer……
Zoloft given to to my daughter in 1999 or so, then removed in 2001 at age 13 due t o increased suicidal talk and horrible agitation, caused 2 months of increased ALL THAT in withdrawals! It wasnt even approved for use in kids then and NO ONE would listen to me until I read out loud, over the phone to the doctor the page in Populous’ bipolar child book abt SSRI induced reactions. It was that one doctor who finally heard me, and any doctor who trialed SSRIs inpatient after that all found out the same thing even though I had warned them of the reaction being guaranteed.
Why keep repeating this? because if those KOLs had not written the PAxil 329 and told working doctors that it was safe for use in teens, then how many teens might be alive today or not have had to endure such awful SSRI side effects?
In 1999 my daughter had a classmate rx Paxil for social anxiety and that resulted in that 11 yr old standing on a cliff edge threatening to jump w/out any prior behaviors like that.
Prescription meds like these are creating life time customers and mental health patients because this is how the med cocktail roulette happens….take one, react to one, cover up that reaction with another, suffer side effects that mimic symptoms in withdrawals, doc increases one or replaces w another….vicious cycle that can be never ending, until frankly, some adults flush the pills and go cold turkey to rid themselves of the med roulette, and find their way by asking themselves about their own life and situations and taking another route that in non invasive….unmedicated therapy, etc.
Also, SSRIs cause this reaction and withdrawal in my daughter and BuSpar, Zyprexa and Abilify did too. It is not always one class of meds, ppl need to watch out for this.
Read the side effects in the tiny fine print of all psych meds and their are psychiatric side effects that most ppl don’t know can happen and then docs medicate THOSE too.
The so-called ‘med washes’ that many child-teen psychiatrists do to kids in the summertime to change meds is not enough time, you cannot simplify med removal into a few weeks and never more than one med together, or you’re asking for a withdrawal nightmare from hell.