C.A.T.I.E. [published in 2005]
Four Common Antipsychotic Drugs Found to Lack Safety and Effectiveness in Older Adults
UCSD News Release
11/27/2012In older adults, antipsychotic drugs are commonly prescribed off-label for a number of disorders outside of their Food and Drug Administration [FDA]-approved indications – schizophrenia and bipolar disorder. The largest number of antipsychotic prescriptions in older adults is for behavioral disturbances associated with dementia, some of which carry FDA warnings on prescription information for these drugs.
In a new study – led by researchers at the University of California, San Diego School of Medicine, Stanford University and the University of Iowa, and funded by the National Institute of Mental Health – four of the antipsychotics most commonly prescribed off label for use in patients over 40 were found to lack both safety and effectiveness. The results will be published November 27 in The Journal of Clinical Psychiatry.
The study looked at four atypical antipsychotics [AAPs] – aripiprazole [Abilify], olanzapine [Zyprexa], quetiapine [Seroquel], and risperidone [Risperdal] – in 332 patients over the age of 40 diagnosed with psychosis associated with schizophrenia, mood disorders, PTSD, or dementia.
“Our study suggests that off-label use of these drugs in older people should be short-term, and undertaken with caution,” said Dilip V. Jeste, MD, Estelle and Edgar Levi Chair in Aging, Distinguished Professor of Psychiatry and Neurosciences, and director of the Stein Institute for Research on Aging at UC San Diego.
Results of the five-year study led by Jeste, who is also current president of the American Psychiatric Association [which was not involved in this research], showed that within one year of treatment, one-third of the patients enrolled in the study developed metabolic syndrome [medical disorders that can increase the risk of cardiovascular disease or diabetes]. Within two years, nearly a quarter of the patients developed serious adverse effects and just over half developed non-serious adverse effects…
“When these medications are used off-label, they should be given in low dosages and for short durations, and their side effects monitored closely,” said Jeste. “Clearly, there is also a critical need to develop and test new interventions that are safe and effective in older people with psychotic disorders.”
Comparison of Longer-Term Safety and Effectiveness of 4 Atypical Antipsychotics in Patients Over Age 40: A Trial Using Equipoise-Stratified Randomization
by Hua Jin, MD; Pei-an Betty Shih, PhD; Shahrokh Golshan, PhD; Sunder Mudaliar, MD; Robert Henry, MD; Danielle K. Glorioso, MSW; Stephan Arndt, PhD; Helena C. Kraemer, PhD; and Dilip V. Jeste, MD
Journal of Clinical Psychiatry. Online: November 27, 2012 [doi:10.4088/JCP.12m08001].
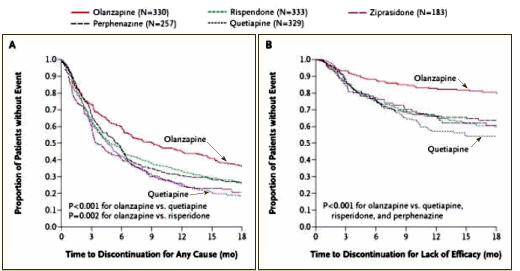
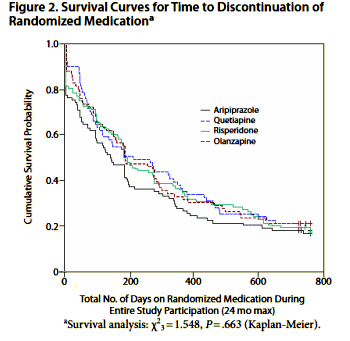
Objective: To compare longer-term safety and effectiveness of the 4 most commonly used atypical antipsychotics [aripiprazole, olanzapine, quetiapine, and risperidone] in 332 patients, aged > 40 years, having psychosis associated with schizophrenia, mood disorders, posttraumatic stress disorder, or dementia, diagnosed using DSM-IV-TR criteria.Method: We used equipoise-stratified randomization [a hybrid of complete randomization and clinician’s choice methods] that allowed patients or their treating psychiatrists to exclude 1 or 2 of the study atypical antipsychotics due to past experience or anticipated risk. Patients were followed for up to 2 years, with assessments at baseline, 6 weeks, 12 weeks, and every 12 weeks thereafter. Medications were administered employing open-label design and flexible dosages, but with blind raters. The study was conducted from October 2005 to October 2010.Outcome Measures: Primary metabolic markers [body mass index, blood pressure, fasting blood glucose, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, and triglycerides], percentage of patients who stay on the randomly assigned atypical antipsychotic for at least 6 months, psychopathology, percentage of patients who develop metabolic syndrome, and percentage of patients who develop serious and nonserious adverse events.Results: Because of a high incidence of serious adverse events, quetiapine was discontinued midway through the trial. There were significant differences among patients willing to be randomized to different atypical antipsychotics [P < .01], suggesting that treating clinicians tended to exclude olanzapine and prefer aripiprazole as one of the possible choices in patients with metabolic problems. Yet, the atypical antipsychotic groups did not differ in longitudinal changes in metabolic parameters or on most other outcome measures. Overall results suggested a high discontinuation rate [median duration 26 weeks prior to discontinuation], lack of significant improvement in psychopathology, and high cumulative incidence of metabolic syndrome [36.5% in 1 year] and of serious [23.7%] and nonserious [50.8%] adverse events for all atypical antipsychotics in the study.Conclusions: Employing a study design that closely mimicked clinical practice, we found a lack of effectiveness and a high incidence of side effects with 4 commonly prescribed atypical antipsychotics across diagnostic groups in patients over age 40, with relatively few differences among the drugs. Caution in the use of these drugs is warranted in middle-aged and older patients.
[Recolored for Clarity]
FYI: Medco is taking it upon itself to redefine what meds can be prescribed and what dose can be written, per my ongoing battle for using Seroquel at 50mg for a diagnosis of Mood Disorder NOS. Absolutely absurd what they forwarded in their denial of paying for the script. Hey, anyone know of literature that documents an FDA approval of Seroquel to treat OCD? I’d truly be interested to know of it, ’cause I haven’t read it anywhere. Yet Medco claims I need to diagnose the patient with this to legitimize a script at this dose.
Pathetic and liable at the same time!
Dr Hassman, for years you and I harped on and on about the potential addictive qualities that Seroquel (antipsychotic for Mood Disorder NOS?) has, and we both agreed the drug should have a controlled substance category added. What surprises me is that you are using it as a broad use med… what may I ask caused you to change your mind/rx habit?
It is interesting that aripirazole had problems with metabolic side effects. I have been skeptical of the claims that it is metabolically neutral based on my experience.
I am glad you have highlighted some of the problems with the antipsychotic drugs. There has been as much hype in this area as with the antidepressants.
After CATIE there was a tremendous amount of spin to recast the findings in a way that continued to suggest the newer antipsychotics were better. Now that they are almost all generic, it is interesting that there is a renewed focus on long acting injectables. Oh yeah, they are still on patent.
Per Stephanie’s comment above, while I am not a fan of Seroquel use broadly, I have had some success with it as low dose, like 50-100mg as above example, so not clear to the question. In the case I am noting, who is it for Medco to refuse a Rx that is low dose and low quantity alone? That should be a concerning intrusion to you folks. Oh, I can write for 200mg as a starting dose, for a Dx NOT cleared in my profession I am privy to now!?
I still would like to see Seroquel be a cntl’d substance, but not so it can’t be accessed. Speaking of controlled substance issues, I will be writing a strong post about benzo’s next week for any interested in reading it at my blog.
Per Dr Hassman’s Medco issue–I do find it alarming that most all of the reasons for dx approval are off-label, esp OCD and insomnia, unless something has changed last I looked those aren’t approved for use, except under doctor discretion, though I have only to wonder that Seroquel for OCD would just act as a tranquilzer and not a solution for that disorder.
The lack of a known biology and a marker creates an unfortunate need to reach outside the box with many unknown risks. I offer respect and admiration for patients whose suffering is often invisible and usually lonely. If CDS alleviates some of the suffering and the patient accepts the consequences, I favor enhancing the QoL.
The real controversy that needs blogged about from all angles of mental health providers is the CPT changes pending. Especially from psychiatry though.
Well, I figured I would tune in again. Playing piano in the whore house just got to be too stimulating.
These kinds of studies M posted are frustrating as they use survival as the principle measure of efficacy. There are many reasons why you lose 75% of your sample in a study, You realize that you could not get a drug approved w/ an attrition like that – longitudinal analysis or not. There are no control groups either. I see these EM things and flip the page. For one thing dosage is a big issue when comparing across drugs. Olanzepine was studied at the top of its range, unlike some of the others. Perphenazine is as effective an AP (aside from Clozapine_ as are the rest. If you did 100 studies and did’nt do the mult. corrections you’d get random superiority one over the other – how many times?
Let’s see: who out there can tell us why the survival curves are congruent in patients who are not homogeneously schizophrenic who are allowed to choose their own medicine. 🙂 It’s real simple. However, Jeste is one of the good guys generally but this is not his finest hour. But to say that for sure – I’d have to read the paper, and spend three hours looking at his disclaimers.
For any of you here in practice, the key horrendous issue in Rx w/ Aps is the incidence of tardive dyskinesia. In fact that is the only reason why we searched. (OH and because we wanted to cure the disease?)
I am not persuaded that the later generation APs are without TD but I am persuaded by pristine data – that the incidences of TD, and other EPS are dose / disease / and age related. Some of the SGAs appear to have the edge. And you pay for that – for reasons just becoming clear as they make you fat, diabetic etc.
No free ride. Almost all of us have or are going to get a disease, That makes us go through stages of grief. Then we find – oh crap – we do not get some easy out. You know: something for nothing. But here is a tremendous secret – well known to friends and practitioners of all stripes. “Nothing is the power of placebo.”
No hang on – Placebo (inert pills, inert talk, inert inert) is the most powerful medicine in my world. It either gets you better w/o side effects (I mean very few) OR it kills you. How’s that for a digital outcome. I’d like any of us, who’ve been hurt or misled by bonafide approved medicines to think that through.
Here’s something else to ponder: why do we drive so much. Remember when we all drove the cost of gas down? Why not drive the cost of medicines down? (I can’t wait to hear the answers to that one!) But if you drive the cost of prescription medicines down by going to faith healers – all you’ve done is . . . etc. you get it.
I’d rather draw generalizations from a series of compact controlled studies, than these sprawling uncontrolled studies. If it were not for TD, the first generation APs, at low dose are as good. I do think when tort arises, that position is defensible if you documented it. Great clinicians do that. They generalize from compact details. The two studies CATIE and JESTE are incomparable, as I see it.
If any of you think antipsychotics like Abilify Olanzepine should not be prescribed for any but the most refractory affective disorders – raise your hands. Which sub populations of of psychiatric patients are at most at risk for TD?
Historically have APs ever been used as prophylaxis for BPD? Care to guess? What’s the problem with low dose Thorazine over Olanzepine if you want to get get fat and have voices stop in your head? Don’t answer all at once.
I do not know why smart people – doctors and patients alike don’t simply go back to the tried and true basics. (no – I am not talkin’ about speaking in tongues.) And then those who are smart, need to share knowledge wit family and friends.
You see what I just wrote. This is what you get when Pharma is not sponsoring the writing, or NIMH is not trying to get public funding for another year. These are a series of generalizations flowing from basic knowledge gained through compact studies, and clinical experience. I’m thinking physicians have to go back to that. CMEs are fine w/ a grain of salt.
But how do we know what is real and what is not?
Given the age of generics now upon us, the issue of big Pharma candy and academic greed will revert to academic greed and snake oil medicine greed. That’s when this blog will hit #1.
I am praying for breakthroughs, not blockbusters. Cancer, Alzheimer’s, Rerouting the bad connections in schizophrenic brain, clear ID of disease caused by vinyl flooring and a non corrupt regulation surrounding it.
I feel good!!
I am currently studying what is to be part of an angry and hurt audience. Please fire at will, with all you’ve got.
BTW except for the specifics, this is not directed towards 1BoM. I have read all of his posts over the past couple of years, and know the guy is brilliant, EXCEL and PP handy and will absolutely gain a following here that will make a difference. Maybe even some of those KOL will stop by for a cameo or two if I keep this up. Come on – you F’kers. 🙂
Regards,
Mark
Don Lalo said
November 29, 2012 | 7:04 pm
“The lack of a known biology and a marker creates an unfortunate need to reach outside the box with many unknown risks. I offer respect and admiration for patients whose suffering is often invisible and usually lonely. If CDS alleviates some of the suffering and the patient accepts the consequences, I favor enhancing the QoL.”
There is my ideal doctor. Maybe my BS meter needs a new battery, too, but I don’t think so. What am I hearing? – Reality. Compassion, Empathy. This is a doctor who will prescribe cautiously, and feel as hurt as his patient if his patient gets unintentionally hurt by the med he prescribed – even if off label. He also knows the risks and the BS. Probably trusts his brain as much as his heart. Excellent.
Before I leave the premises:
We are in doo doo because the FDA does not – unless they do now – generally allow comparison labeling. If it did, considering the use fees they charge, it would make a difference. I think a big one. To do a comparison trial you’d need to study homogenous populations at a range of doses for each drug +/- placebo (I have to think about that) – yes Placebo. Now Jay and others will tell you that 7 arm studies are tough and expensive. But wow! not perfect; but wow. They are really the only post marketing studies that should be done for EBM. Sure it will eat up their profits. But not enough to stop innovation, especially if tort reform comes about in some measure. (another can of worms!) What do you think?
OK one more. That’s it. Promise.
Dr. Hassman said ; ” FYI: Medco is taking it upon itself to redefine what meds can be prescribed and what dose can be written, per my ongoing battle for using Seroquel at 50mg for a diagnosis of Mood Disorder NOS. Absolutely absurd what they forwarded in their denial of paying for the script. Hey, anyone know of literature that documents an FDA approval of Seroquel to treat OCD? I’d truly be interested to know of it, ’cause I haven’t read it anywhere. Yet Medco claims I need to diagnose the patient with this to legitimize a script at this dose. Pathetic and liable at the same time!”
Medco was sold to Express Scripts; I expect this kind of thing.
Anyway it is pathetic and so is rampant OCD – especially if it is of the severe non-comorbid variety. No. SRQL has not been approved for OCD. But OCD is such a challenge to Rx by any means. Forget PBO. In order to discourage off label use, they are begging you to change your Dx to BPD/schizaffec and prescribe the equivalent of 6x the min effective dose for BPD/. It can only be made practical by giving the patient separate written dosing instructions and documenting exactly what you are doing and why. See, that opens up a can of worms does it not? Are there cases in wh/ the insurer has been held liable for this kind of thing? Given the relative subjectivity of clinical psychiatric Dx, does the patient have the criteria for the change in Dx?
I guess this is related to the graphs M posted. If is came to blow, I suppose you’d have to use the bogus papers by some KOLs to support your case? 🙂 I’m smiling – but I know the seriousness of having your hands tied, when you are well intentioned, and see the need for your patient and their finances.
Do you have anything for 1 Boring Old Man Blog Addiction. Forget it, Dr. I have a piano to play in a whore house. I am now late for the gig. Geez!
Good luck!
‘Where does piano player in a whorehouse mean?
A piano player in a whorehouse is one that claims innocence in an unethical situation.
To what extent does the piano player contribute to the illegal goings on around him?
It is a statement that implies that the metaphorical piano player has a mitigated culpability.
It could also mean that the piano player is superfluous; i.e. People don’t go to the whorehouse to hear the music.”
I believe both definitions are appropriately applied in this particular case!….Yet, your self reflective rendition of this following touching ballad dedicated to Charlie, drew rave reviews from head Mistress http://www.youtube.com/watch?v=jn0A1ExLupw 🙂
For those of you who cannot read the signs, the prescription of atypical antipsychotics has peaked. Risks and lack of benefit are being more widely acknowledged.
Now all you need to do is face the patients you have harmed. How would you like them to contact you?
Nicely clever. 🙂 I liked your creativity (unfortunately at my expese – stealing ) re the metaphors. They took work. Why not contact me off line “Barney” if you are concerned about confidentiality? Seriously. contact support@thejazzmall.com and it will probably reach me. Do you like jazz? If not, understandable.
I try to close w/ humor whenever I feel it. My aim is neither to humiliate you or to call you off-handed names. Because the internet is forever, and b/c I am posting as myself – what you are doing is like what a sniper does. I am not even distantly related – i think, anyway (wait are you my wife??) – to the cause of your attacks. This I know.
So, that is why I can only assume that you have to be hurting. You’ve got me on trial for being me. It’s been many years – but isn’t that transference? If so you are a secret admirer? 🙂 Wow.
Charlie Nemeroff. Again just because I’ve known him, and to make matters much worse w/ you , that he tried to support me many years ago b4 this stuff came down, that does not mean I am an enabler, or supporting him . . I’ve spent a lot of time pondering the notion of appeasement, diplomacy, strength, and actually self absorpton. I’d love to talk w/ you about that off-topic off line if you are open to it. Also there are the heavy issues of morality and culpability you broached.
Yes, I am the fool -here – and most places at the times I venture forth. Blame Jay if you know him. It takes a lot of work to be a professional fool. Feel any better? When I say I am a fool – well that has meaning. (take a look at the role of the fool in really problematic situations: this one included ) However, when another says I am a fool – without even offering a tip, as the King would do – it simply signifies a person who punches vicariously from the sidelines. It’s by the tip jar a FOOL knows who is the KING and who is – a frstrated KING. Cool, Huh? See,you can enjoy what I just said, and not feel weak, or lonely.
So. If this b-Log stays open you might – Barney – decide to fire back some more bombs. Otherwise you could simply accord respect. What you will do is interesting to me. You will not lose face here if you decide to go the civil route. Whatever you do I’ve invited you to contact me personally. You may have some good idea on how to proceed.
Be well.
Mark
P.S. The whore house was one of the places that supported the formation of jazz. The links that you posted – those tunes owe equal influence to jazz and country – wh are very related. Having played actual brothels and strip joints as a young guy needinng to make money while in school, it was fun. The music was respectfully/attentively received by many. The women who worked there were OK (actually pretty indigent) , and so was the Mr. Madame as I recall. I wouldn’t equate that old business with KOLS and jerryrigged publications. Long ago, before sports TVs on the walls, these brothels competed w/ each other by hiring pianists who could really play. There wee competitions. So when I joke I am going to the brothel to play – it is more wishful thinking than not. See today, they do have those TVs on the wall, and hardly anybody listens. I donlt get that, b/c I’ve been at this for 52 years. So, unless it is w/ friends I do wait for the overseas gigs, or operas, or concerts at universitites. Ther ar some here who’ve attended. But “Barney” I am the only one on a major record label have published in Science, and lecturedat Nobel Hall. I do feell proud of those objective things. But i shows you that I feel vulnerable too. How could I be a KOL like Charlie we/ that kind of thin skin Yu won;t beieve it but these things are fact and I like to say them because they make me feel like somebody. But deep down, we are all pretenders. And if you could be real – I would tell you why I think that is so. OK? Why do you think my music is bastardized versions of songs? I’m interested.
With whom is Altastrata arguing and why?
While some ponder and read the comments don’t forget that there is voting for the pharma-dream-use-more-pills-give-more-dx-for-profit DSM5 tomorrow
http://www.npr.org/blogs/health/2012/11/30/166252201/weekend-vote-will-bring-controversial-changes-to-psychiatrists-bible
Listen at NPR link in the article
Does not the piano player in the whorehouse consider himself morally superior to the working girls? He takes no responsibility for what goes on outside the parlor.
Mark Kramer, in another thread you maintained there was some good data in pharma-sponsored studies. Dr. Mickey has likened finding that good data to cleaning out the Augean stables. If you know which studies are reliable, you would be doing psychiatry an immense service by publicly identifying them.
Why not take up stable-cleaning rather than piano-playing?
Is this thread making subtle reference to “whores and cowards” that has been used to describe the mentality in the profession of late?
My take on this study:
http://www.madinamerica.com/2012/12/a-recent-study-of-atypical-neuroleptics-the-results-of-our-study-are-sobering/
Hi Sandra –
I read your link.
It’s interesting but Dilip’s main contribution is to remind us of this:
” . . . we should be very careful in using any of these drugs in people over 40, especially if we are using them off label for any length of time.”
Well, of course! Again the devil is in the details. These drugs are there for us to prescribe when milk and cookies do not work. They can be particularly helpful starting at the low dose intermittently for the elderly (which in most circles means aged > 64 years) i.e. , acute agitation in people w/ dementia. Don’t you agree? As you know these may help in the short run, occasionally long run, so that the patient can convalesce from other medical procedures, etc. My impression is that these drugs may be prescribed too often for a kind of chronic near chemical restraint off label which would not be across all mental disorders universally if ever appropriate. At issue are not his general conclusions. Jeste has always been a thoughtful man. He offers basic politically correct clinical pablum and common sense.
However, a critique of the science behind Jeste’s paper is required – those issues of design which may or may not support each conclusion. If you have the full article, could you email it to me through 1BOM?
One needs to look at the randomization scheme in terms of reasons for patient and doctor wanting to switch (safety/ inefficacy/ quantifiable pathology etc.), whether the patients who accepted entry had ever received any or which of the drugs offered at randomization, how many patients may have received blinded medication on which they already failed in terms of efficacy or safety, whether patients were stratified in the randomization according to Dx and age.
The outcome of this study is not typical in that others (of any sponsorship) have shown that merely switching medications in homogeneous marginally responsive populations does generally bring about some improvement in quantifiable psychopatholgy . Also in such a diverse sample one would want to see not just a BPRS or PANSS but items and subfactors as hypothesis generating clues for compact studies. THe problem w/ the study is that it does not offer any specific guidelines except “don’t over do it!” or “don’t do it” Tell that to the clinician whose patient is in a rehabilitation unit following a fall and fracture – is disoriented, combative, and referentially deluded. And in whom Milk and Cookies, family, and great support are ineffective.
If this were a study of 4 brands of regular gasoline in 6 different types of automobiles (one being diesel , another electric, and a third a hybrid, the others standard but different models ) , and the primary outcome was time to drop out of the study, and the secondary was general engine performance . . . . well, take it from there Doctor. ROTFL as they say.
On the flip side it is the kind of research if taken literally w/ its potential design flaws wh can be taken to support a soteraist philosophy – which has its place, when it does – but can be a disservice when it fails.
Regards, Mark
Hi Altostrata (HIGHROAD?)
The working girls were great friends. As you know, there is a huge difference between being discriminating and being judgmental. If ever one is judgmental keep in mind 1FF3FB (1 finger forwards, 3 back.) Be careful. Those who are the consumer advocates attempting to protect the general population from you know all those ignorant evil doctors and deceptive promotional practices – running blogs and accepting ads or donations from alt. therapies . . . you are on the edge of C.O.I. You should grab the name bassostrata while it still is available.
I really hope that you are not suggesting that people are immoral if they do not give up their passions in order to clean the crap out Psychiatry’s stables . If so that is either really quite mean (controlling, judgmental, and meant to humiliate a beastly Daddy or Mommy ); or that you really do understand the story of Heracles and the Augean stables and urge all to that – if they should so wish. People who follow their dreams are neither the problem nor the enemy; they nearly always are the solution.
I aspire to a Heracles – always have and always will. His work not done through blogging. Blogging – is taking a rest – taking a dump – not work. Heracles – who for all we know may have been a decent musician – his work is done through diversion of very powerful forces, not his powerful force: diverting 2 great rivers so that they can purify whatever needs purification. The two rivers which are currently and which always have been in my awareness are the river of Innovation, and the river of Understanding. In Science there is a great third River: Serendipity. Sooner than we would think change occurs. Like Now.
The end of the Heracles story is not appetizing. It’s nothing to be advocated. Also the stables do stink just now, especially badly, to those who whose primary agenda is to widen vegetarian market-share.
You can’t understand the show completely until you visit backstage and then the basement. This goes for anything that seems to glitter.
Regards, M
“Is this thread making subtle reference to “whores and cowards” that has been used to describe the mentality in the profession of late?”
No – just a new member of this forum.
Stephany –
I am not a fan of the endless DSMs for many reasons. The originals made sense when they were attempting to dissect what appeared could be dissected on the clear differences at the time between Lithium,Chlorpormazine and Tofranil. vs. the overgeneralizations of psychoanalysis towards “mental illnesses.”
Yes it may now be a boldfaced publishing business, I think.
M
Yes, I agree it’s about profit in the publishing business…