GlaxoSmithKline doesn’t seem to be able to kick the bribery habitThe GuardianJuly 28, 2013via PharmagossipThe last time anyone remembers a row over Brits forcing drugs on the Chinese market, it ended with our boys taking Hong Kong. But anyone predicting that British interests will emerge from the latest China crisis with comparable benefits to those of the opium wars has surely been smoking some pretty muscular gear. To recap: last week, UK pharma firm GlaxoSmithKline admitted that Chinese doctors were bribed by its execs with cash and sexual favours in return for prescribing the company’s drugs. That coincided with rival AstraZeneca having its Shanghai office raided by police – all of which is jolly inconvenient, as Astra faces the City this week to unveil its interim results.
Some investors ponder whether bribery is a wider problem than has yet emerged, and if Chinese authorities are deliberately targeting foreign firms. Maybe, but critics of the UK companies also point to GSK’s $3bn fine last year for bribing US doctors, plus Astra’s indictment in Serbia on similar charges, as well as an admission in its annual report about "investigating indications of inappropriate conduct in certain countries, including China".
All of which leaves one obvious question: when it comes to getting embroiled in bribery probes, is British pharma struggling to kick the habit?
Forbesby Robert RadickJuly 28, 2013also via Pharmagossip
When it comes to allegations of foreign bribery, what a difference a week can make. Just over ten days ago, a new player in the world of international bribery – the Chinese Ministry of Public Security – announced that it was investigating senior executives in the Chinese division of British pharmaceutical company GlaxoSmithKline for allegedly offering bribes to officials and doctors in order to boost company sales. GSK responded at the time with the respect and deference that one would anticipate, stating that it takes “all allegations of bribery and corruption seriously” and that it would fully cooperate with the Chinese authorities. Regarding the merits of the allegations, however, GSK stated that the company had already conducted an investigation and “found no evidence of bribery or corruption of doctors or government officials.” Now, in a story that continues to evolve rapidly, GSK appears to have acknowledged the misconduct that the Chinese authorities alleged…<the article catalogs the details of the tumultuous week for GSK in China>.
Like Alex Gorsky at Johnson & Johnson, Andrew Witty joined Glaxo as a young man, and worked his way up through the company ranks to finally become CEO of GlaxoSmithKline [being Knighted in the process]. And like Gorsky, he’s been along for the whole ride intimately involved in their marketing strategies ["He served as a Vice President and General Manager of Marketing of Glaxo Wellcome Inc., a subsidiary of GlaxoSmithKline with responsibility for strategy development, marketing execution and new product positioning. He served as an economic adviser to the Governor of Guangzhou, China from 2000-02."]. It’s a bit hard to hear him blame "‘individuals’ who operated outside ‘controls and processes’," as if this business in China is some kind of big surprise to anyone. He’s been CEO since 2008, and the Chinese say this scandal started around that time.
"All of which leaves one obvious question: when it comes to getting embroiled in bribery probes, is British pharma struggling to kick the habit?"
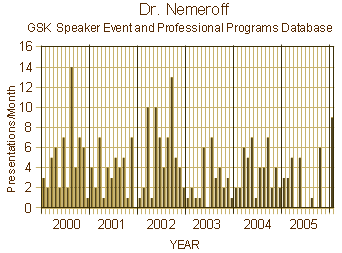 Personally, I don’t see them having any moral "struggle." Sticking with the addiction metaphor, it looks to me a lot like the "denial" seen in addiction. Witty is explaining that they have some rogue executives in China – arguing that this is a special case rather than a barometer measuring the pulse of GSK’s overall marketing program. Were Study 329 and Study 352 special cases? Was sending toxic Charlie Nemeroff on the road hawking their wares a special case? How about the ghost-written textbook [Recognition and Treatment of Psychiatric Disorders: A Psychopharmacology Handbook for Primary Care] or the generous collaborative grant with the NIMH to Dr. Nemeroff yielding nothing of scientific interest? While some of these examples came to Glaxo when they acquired SmithKlineBeecham, it was SKB’s marketing successes they bought – and they picked up the ball and ran with it.
Personally, I don’t see them having any moral "struggle." Sticking with the addiction metaphor, it looks to me a lot like the "denial" seen in addiction. Witty is explaining that they have some rogue executives in China – arguing that this is a special case rather than a barometer measuring the pulse of GSK’s overall marketing program. Were Study 329 and Study 352 special cases? Was sending toxic Charlie Nemeroff on the road hawking their wares a special case? How about the ghost-written textbook [Recognition and Treatment of Psychiatric Disorders: A Psychopharmacology Handbook for Primary Care] or the generous collaborative grant with the NIMH to Dr. Nemeroff yielding nothing of scientific interest? While some of these examples came to Glaxo when they acquired SmithKlineBeecham, it was SKB’s marketing successes they bought – and they picked up the ball and ran with it.
…based on the aggressive pursuit of the GSK matter by the Chinese authorities, overseas executives are forewarned that other sovereigns – including those in countries that lack a well-established tradition of civil liberties and due process – may be actively staking out their own territory in the fight against bribery and corruption…
For overseas executives in foreign countries with a tradition of corruption and bribery, there was already ample cause for concern that such issues might ensnare them in law enforcement investigations. But until now, there may perhaps have been some measure of comfort from the fact that such investigations traditionally arose out of the United States or the U.K., where the criminal process necessarily involves the filing of formal charges, bail hearings, and other hallmarks of due process. Overseas executives may even have held out hope that if troubles arose with overseas governments, their employers would bring them out of their foreign locales and back to their country of origin. Now, overseas executives accused of bribery need to take seriously the prospect of travel bans, summary arrest, and detention in foreign prisons…
For the pharmaceutical industry, attacks from the medical community and other commentators stem from three sources not acknowledged elsewhere. First, at the start of themodern era of medical practice, in the 1950s, the basis on which drugs are patented was changed, allowing companies to have monopolies on drugs in a way they hadn’t had before. This permitted the development of blockbuster drugs, laying the basis for the marketing of drugs we now see. Second, also in the 1950s these drugs were made available on a prescription-only basis, putting a relatively small group of people with no training in or awareness of marketing techniques – doctors – in the gun sights of the most sophisticated marketing machinery on the planet. Third, having been under siege by industry for a century, in the 1960s we in medicine woke up to find a horse outside the gates of Troy in the shape of controlled trials and hauled it inside, not realizing that industry designed trials provide wonderful evidence about the benefits of drugs but very little evidence of what might be best for patients.
Against this background, discussions of conflict of interest and ghostwriting, while important, only scratch the surface. We need to ask whether faith in controlled trials when we are not allowed to see the data from those trials can protect us against the biases introduced by commerce. Whether making drugs available on prescription-only status is appropriate in an age of blockbuster drugs. Whether the basis on which we now permit drugs to be patented is delivering the benefits to society that patents were originally designed to provide. As I will argue, every assault on industry paradoxically has instead reinforced the apparent need for controlled trials, prescription-only privileges, product patents, and further regulatory arrangements, and together these have bound the medical profession and government even tighter to the pharmaceutical industry. These are changes that among them laid the basis for companies to create blockbusters, and as long as these changes remain unquestioned, industry is happy to settle back on the ropes and take a pounding…

On a related note this may be of interest: Surgeons Eyed Over Deals With Medical-Device Makers July 26, 2013 Wall Street Journal print edition pg. A1 and A 10.
This is a story often heard with a doctor becoming a distributor of a medical device whose surgery and usage rate jumps remarkable along with the resulting increase in malpractice suits and countless people left in pain and in need of additional surgery.
What happens? The usual, the doctor leaves the practice before his license is revoked and sets up shop half way around the country. There is intimidation and denial along with shifting the blame to all of those who are blameless.
This appears to be the standard in medicine: Denial, shift the blame, and never be responsible for all of the pain and suffering, all the while cashing big checks at the bank.
Steve Lucas
Hello,
My name is Joshua Aldridge. In 2008, I received a BA in psychology from Georgia State University, but set it aside to pursue fiction writing. I sold furniture, managed a RiteAid and catered weddings to support myself as I worked on my short stories and novel. When I failed to make any notable headway, I cut ties with everyone I knew and moved back home. Initially, the asceticism hardened my resolve, but after a while my thoughts and writing habits became obsessive and circuitous. I’d move sentences back and forth for weeks, convinced of the incremental gains I was making. Whatever narrative I’d begun with inevitably fractured into dry exercises in technique. It’s been five years now and I’ve come to understand I’m not a fiction writer, but I think I could make a good scientist. I got back into psychology late last year and have been reading your blog since. I’ve been studying for the GRE, but I also need three recommendation letters. I don’t expect to get one on the merit of a single comment. I’m not working many hours right now and can do editorial or secretarial work. Whatever’s needed. I’ve a pretty flexible skill set.
Thanks for your time, and for the blog. It’s a great read!
There seems to be a recurring message, albeit small and limited so far, to justify that sociopathy in fact is BENEFICIAL to the human existence as of now. Yeah, just what we need, people out there running large influential businesses or maintaining a place in state or federal politics, who have NO clue probably even how to pronounce the word “empathy” much less define or relate to it’s intent, and believe that the needs of the one or small circle of few others around said one are much more important than the needs of the many.
And you know what, these fine examples of greed, lust, and lack of accountability keep getting swept under the rug and then stood on top to be ignored, rationalized, minimized, or just pathologically defended as “it is what it is”. As long as there is a sizeable majority of people who not only accept the false premise of “biochemical imbalance” or “better living through chemistry”, but demand it, nothing will change.
And fining these companies or individuals is useless with the monies being made. No, as I have said over and over, find creative but applicable felony charges against specific individuals that really count in the public opinion process, and watch these people squirm, as much as their counterparts who haven’t been caught yet.
Sociopaths as a whole don’t like prison. But, it is an environment that is basically devoid of empathy, so why shouldn’t they instinctively just fit it?