[Note: Dates corrected] Back in January 2012, I wrote about Australian Depression/Sleep Guru, Ian Hickie, who had published an article in the August 2011 Lancet reviewing the studies of Agomelatine as an antidepressant positively [Novel melatonin-based therapies: potential advances in the treatment of major depression] to an international outcry of protest [of sound and fury…, it’s about time…]. His petulant, near paranoid, response to the criticism came to nothing. Now there’s a new meta-analysis that sets the record straight – and more:
by Markus Koesters, Giuseppe Guaiana, Andrea Cipriani, Thomas Becker, and Corrado BarbuiThe British Journal of Psychiatry. 2013 203:179-18.
Background: Agomelatine is a novel antidepressant drug with narrative, non-systematic reviews making claims of efficacy.Aims: The present study systematically reviewed published and unpublished evidence of the acute and long-term efficacy and acceptability of agomelatine compared with placebo in the treatment of major depression.Method: Randomised controlled trials comparing agomelatine with placebo in the treatment of unipolar major depression were systematically reviewed. Primary outcomes were (a) Hamilton Rating Scale for Depression (HRSD) score at the end of treatment (short-term studies) and (b) number of relapses (long-term studies).Results: Meta-analyses included 10 acute-phase and 3 relapse prevention studies. Seven of the included studies were unpublished. Acute treatment with agomelatine was associated with a statistically significant superiority over placebo of –1.51 HRSD points (99% CI –2.29 to –0.73, nine studies). Data extracted from three relapse prevention studies failed to show significant effects of agomelatine over placebo (relative risk 0.78, 99% CI 0.41–1.48). Secondary efficacy analyses showed a significant advantage of agomelatine over placebo in terms of response (with no effect for remission). None of the negative trials were published and conflicting results between published and unpublished studies were observed.Conclusions: We found evidence suggesting that a clinically important difference between agomelatine and placebo in patients with unipolar major depression is unlikely. There was evidence of substantial publication bias.
Unfortunately, the article is behind the paywall, but if you’re interested in the emerging standards for systematic meta-analysis, this would be a good value for dollar addition to your collection. My thought on reading it was that science has finally arrived to the world of CNS drug clinical trials. I had a rookie’s stab at analyzing Hickie’s review in January 2012 [of sound and fury…], but this is the definitive version. They included unpublished studies, assessed the articles for publication bias, and computed a comparative index of strength of effect for the various trials [expressed as mean change in HAM-D Scores]. 9 of the 10 studies with HAM-D scores were included. I took the values for mean, stdev, and n for these 9 studies [only 5 of which were published] from figure 2 in the paper to illustrate several points revealed in this kind of systematic meta-analysis.
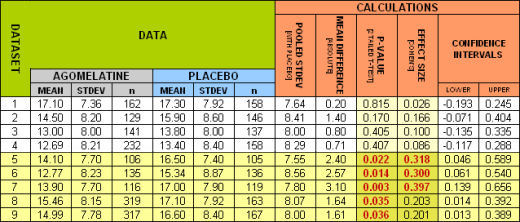
The lower five studies were published. Notice that they were the only ones where Agomelatine separated from Placebo [p < 0.05]. The Effect Size [Cohen’s d] is a measure of the strength of the drug’s effect. While there’s no fixed value for this parameter, 0.25 is a usual cut-off. Notice below which studies got published and which didn’t [shown with the 95% Confidence Intervals]. The diamond shapes represent the weighted subtotals:
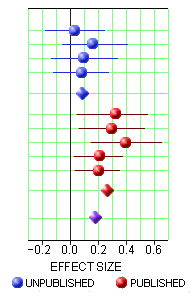
And finally, the graph known as a funnel plot predicts that the smaller the number of subjects studied, the more variability there will be in the results:
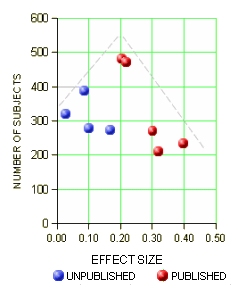
As you can see, publishing only the more desirable of the smaller studies gives a falsely inflated impression of the drug’s effectiveness. They aren’t better, just part of an expected variability. In the paper, the authors’ make a vitally important point about drug approval:
Problems with current methods for approving new drugs
In Europe new drugs are approved or rejected on the basis of the results of studies carried out by the manufacturer and submitted to the EMA. We note that decisions are taken on the basis of the results of individual studies with no role for aggregating efficacy data using meta-analytic techniques. We argue, however, that pooling studies would have some beneficial consequences for the review process by increasing statistical power and by contributing to the detection of between-study heterogeneity.
I find it ironic that the rigorous scientific technique that we’ve needed to evaluate the CNS drugs that have flowed out of the mythic pipeline for the last quarter century are now finally being published in the form of independent meta-analyses like the one in this article. The irony is that the real techniques have been refined to debunk the sea of bad and misleading articles and analyses that we’ve endured for such a long time. Would that they had been applied the first time around! Nevertheless, they are a breath of fresh air and long overdue. My compliments to the authors. Oh yeah, Dr. Hickie, Agomelatine is a dud!
The general question I want to ask is this: how relevant is, for a psychiatrist’s job, his/her medical training? And I don’t say this as a joke.
After many entries, comments, etc, most people seem to agree that what psychiatry calls “diseases” are not such in the biological sense, just patterns of behavior that some self appointed “doctors” call pathological. Would it make any difference if that determination was made by self appointed statisticians or data scientists? Is it the insistence on having doctors make that determination only a way of improving the “placebo effect” since in many people’s minds “medicine” is the new “God”, and therefore “doctors” the new “priests”?
Wouldn’t we all we better off if at least the new priests recognized what their true role, and value, truly us?
Cannot,
Wow, I think we could write a book about that comment. First, and I am not a doctor, I do believe that some mental health problems originate in some biological issue as well as any number of traumas. That said, I do not believe that all mental health issues can be resolved through the use of medication alone. Medical knowledge can be of great use in determining the proper course of action for these folks.
Secondly, I do believe that no matter, biological or behavioral, everyone facing a mental health issue can benefit from time spent with a trained mental health provider. Friends my age talk openly about how Dad would stop at the bar, have this tiny beer, unload about the boss, wife, and kids and then go home kiss the wife, pat the kids on the head, and talk about their great day at work. This chance to openly get rid of all this pent up frustration is sadly missing from today’s commuter society with 24/7 connectivity and no privacy.
I see the parallels between medicine and the clergy. There is an old saying in church that if you want to see what people feel is important look at their checkbook. Today for many families’ medical care and insurance is their number one expense, exceeding housing, food, transportation, education, and church donations. Churches have adopted the very worse of pharma sales practices while, you are correct, many in medicine have adopted a religious overtone to their practice.
Doctors often expect a certain reverence and obedience while not allowing discussion and opposing opinion.
This only touches the surface of a very thought provoking comment.
Steve Lucas
I’m doing a sleep study in a week. For the second time, I will exercise my right as a patient not to fill out that little inventory to see if I’m “depressed.” I don’t even feel bad, and as far as I’m concerned it is not a legitimate instrument of diagnosis, but a funnel for the pipeline that is polluted with corruption and venality.
I recommend that everyone do this. Those little forms get passed out by all kinds of medical doctors. Rather than jumping onto the greased slide of BigPharma, patients can require doctors, nurses, and other medical professionals to look them in the eye and talk to them and listen.
Steve Lucas,
Thanks for your thought. Indeed, my comment was meant to be “thought provoking” but I mean it 100% :D.
From where I stand, the medical training of psychiatrists is just cosmetic so that they can fulfill their role as this day and age’s behavioral police. Having a “medical degree” makes that role more acceptable to the general public than having a “theological degree”.
The role of the psychiatrist in our society is no different though from the role of the priest when Karl Marx called religion “the opium of the people”. We are past the time when religion plays that role, but surely psychiatrists give literally “opium” to the masses.
Not a bit surprised about agomelatine being the usual psychiatric vaporware. How much longer can this go on???
Interesting. I was in a room with Prof. Hickie just last week, as we were both attending a lecture at the Sydney Brain and Mind institute (of which he is the executive director.
Prof. Hickie is certainly a ‘key opinion leader’ over here in Australia (although in the States you might view him as being a big fish in a small pond, he is certainly a very big fish by Australian standards). He also has links -which he disclosed- to Servier, the manufacturer of the drug he was spruiking.
Anyhow, thought you might be interested to see his rebuttal to the initial criticism that met his first publication. As usual, it’s full of victimization and Ad Hominem, but it’s always interesting to see both sides of the story.
http://www.theaustralian.com.au/news/health-science/campaign-targets-depression-guru/story-e6frg8y6-1226269135293
Thanks Adam. Actually, he’s a pretty big fish in his own right internationally. I was actually just looking through the correspondance in the Lancet following his article. The authors of the meta-analysis were among the first to cry foul. Novel melatonin-based treatments for major depression.. There’s a full thread of correspondance linked on that page.
Adam,
“Professor Hickie is now negotiating a $100,000 research grant with Servier on the effect of its drug Valdoxan on the circadian system but says this was not on the horizon when he wrote The Lancet paper two years ago.”
This seems a copy paste of this hilarious, and Australian, parody of disease mongering https://www.youtube.com/watch?v=RoppJOtRLe4 :D.
It’s nice to see something uplifting, thank you!
Wiley,
As a person with sleep apnea who has had a horrible time adjusting to pap therapy, filling out that questionnaire would be the kiss of death. So thank you for that warning as if I see that form when dealing with sleep professionals, I will definitely refuse to fill it out.
I was speaking loosely, perhaps, AA. I don’t know that they’ll give me an inventory, but the pain nurse I talked to did. She didn’t have any problem with me refusing it. If I am not given the depression questionnaire, I will be very surprised.
Cannot,
The Marx comment seems appropriate given today’s drive to medicate. I am a business person and saw an article a short time ago laying out how a psychiatrist seeing 40 patients a day could generate over a million dollars a year in revenue. This allowed the hospital to pay more than a person would make in private practice and represents that all important revenue center in today’s business model.
I represent the goose that laid the golden egg to doctors as being older and still covered by private insurance. I have had doctors physically block the door of the exam room demanding I agree to additional testing and medications claiming I was; “Taking food off their children’s table.”
Today I find friends who are totally lost. They went to college, had great jobs, borrowed money and when the job was gone and the house value fell they found doctors more than willing to “help them through this” with some type of medication. This was done all within the five to seven minutes of actual time the doctor spent with them in the exam room for physical issues.
We now are forced with no training to ask the question: Is this behavior due to the stress of the situation or a result of medication?
Mickey made two points that were really telling: First, he sees patients where medications now make up a “food group.” Second, the medications have become the justification for the person’s problems. I have seen people use their medications as an excuse to drop any pretense of social norms and engage in behaviors that drive people away when this is the time they are needed the most.
The old pharma battle cry of “ a pill for every person, and every person taking a pill” is truer today than ever before, with the addition of an EMR driving up coding and, as noted, those records never going away we have a situation of never ending medication for far too many of the population.
Steve Lucas
SL, TCR,
Time for another The Onion classic,
http://www.theonion.com/articles/pfizer-kingpin-gunned-down-in-ongoing-prescription,31678/
🙂
Steve, some behavior you describe could be the effect of a drug or drugs affecting cognition.
http://www.frontiersin.org/Integrative_Neuroscience/10.3389/fnint.2013.00067/abstract
Interesting, wiley. SSRIs interfere with learning from unpleasant situations. Is a survival strategy being canceled out so people can tolerate bad marriages and bad jobs?
Steve Lucas said… “Wow, I think we could write a book about that comment.”
Thomas Szasz wrote a few!!!
Altostrata wrote “SSRIs interfere with learning from unpleasant situations. Is a survival strategy being canceled out so people can tolerate bad marriages and bad jobs?”
Of course the masters do not want their slaves to be unhappy. (They may be revolting!) Szasz drew attention to drapetomania for this very reason.
Dr Hickie has disclosed his relationship to Servier in the Lancet paper. However – there is data in the paper that have never been published before, so I conclude that he got unpublished data from Servier, and that is not fully transparent in the paper…