This post has nothing to do with psychiatry, but everything to do with the pharmaceutical world. It’s about the so-called blood thinners, anticoagulants taken to decrease clotting in conditions like atrial fibrillation, a heart rhythm abnormality associated with clot formation that can lead to stroke. The standard drug, Warfarin, has been in use since the 1950s. It interferes with the Vitamin K dependent factors in clotting, so it’s effect can be reversed with Vitamin K. Obviously, the main problem is abnormal bleeding, and so clotting has to be checked frequently.
Comes now Pradaxa®, a new drug that acts on a different part of the clotting mechanism. It doesn’t require the frequent blood tests like Warfarin, at least that’s how it was introduced. Abnormal bleeding is less than Warfarin for certain kinds of bleeding, but perhaps more for gastrointestinal bleeding. There are no simple blood tests to monitor it, and it can’t be rapidly reversed.
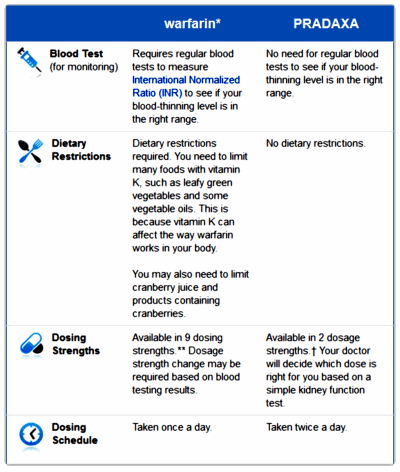
It’s patients suing the drug firm over Pradaxa-related harm that led to the articles in the Times. The judge ordered release of some documents related to the case that include internal company e-mails about a research study coordinated by a company scientist, Paul A. Reilly. Reilly’s study showed that a part of the safety problem with Pradaxa was that some patients had too high a blood level and some patients too low [sound familiar?]. He concluded that a blood test that measured drug levels could be helpful for at least some patients in avoiding dangerous reactions. [Such a blood test is available now in Europe but not the U.S.]A draft of Reilly’s paper that included these findings unleashed a storm of e-mails from other company scientists and officials. They argued that publishing a paper with these conclusions would undermine the company’s primary marketing point in favor of Pradaxa, the result of a fine-tuned marketing effort going back a decade. Moreover, some feared that if the paper were published, it would be that much harder to get the government regulators to hold off demanding blood tests. The end result was that the paper was published recently but with many of the offending details removed.
Boehringer Ingelheim insists that this was a simple matter of scientific review and refinement. A draft was circulated, others chimed in with appropriate criticisms, and in the end the final paper was suitably modified to better present the actual facts. Nobody here but us scientists, boss…

Is it really wanted to publish this exposure event paper of RELY?I cannot believe that for a decade a drug was developed with the clearly defined target of no monitoring needs, a prospective trial without plasma level monitoring was performed generating the RELY study results, that we promote 2 fixed doses without monitoring, defend continuously to Health Authorities that individual patient characteristics do not allow a dose translation based on plasma levels only and then finally release a publication where exposure event relationships which was neither prospectively defined nor adequately conducted are described to define an effective and safe plasma level range…This will make any defense of no monitoring to HA extremely difficult [ie Health Canada, TGA] and undermine our efforts to compete with other NOACs.As I am not empowered to release or stop any publications I would like to ask you to check once again whether this is really wanted.
"Unless we can find a solution to the commercial incompetence problem, we have to recognize that the pharmaceutical industry has an irreducible conflict of interest in relation to the way it represents its drugs, in science and in marketing. And unless we can resolve this in a way that is more in the public interest and in patients’ interest, I would argue that drug companies should not be allowed to evaluate their own products."
“So it may not be directly about psychiatry, but it sure is directly related to psychiatry’s problems…”
Or the other hypothesis is more true – that it is really not specific to psychiatry, but that Big Pharma conflict of interest is present everywhere.
Unlike other medical fields, the practice of psychiatry is almost entirely dependent on the prescription of drugs, most of which have been deliberately and heavily misrepresented for the last 30 years.
I have tried to take to heart the gentility of the previous post. I want to emulate our host. However, I will say that the fact that there are problems throughout medicine does not negate my own criticisms of psychiatry. There may be problems we share with our colleagues but they are problems nonetheless. There may also be problems – due to our diagnostic uncertainty, for instance, – that are particular or at least more problematic for psychiatry.
Sandra,
I agree with that. If anything, I’m a bit dismayed to see medicine “coming our way” as in Statins, as in Pradaxa, as in MRIs and echos galore. But I think we were an easy mark. Subjective. Poor. A target for managed care. Hungry for science. And in academic psychiatry and organized psychiatry, we selected for people who figured out how to follow the money. I hate to be so plebeian about it, but that’s what it looked like in the day. We used to have greats, people we went long distances to hear. Beloved chairmen. Experts of stature. And at a local level, there were respected clinicians who would always figure things out. Now we have KOLs. I don’t think there has ever been the intellectual poverty in mainstream medicine we experience in modern psychiatry.
The proof will be if the American College of Cardiology jumps on this Pradaxa thing and gets to the bottom of it. Academic/Organized psychiatry has not done that through all of our exposes and scandals. They’re either silent or worse, part of the defense. I think that’s why the Pradaxa story caught my eye. I realized that my expectation was that Medicine proper would get involved in an active way. Psychiatry hasn’t done that.
So much for my gentility, or maybe my gentility is reserved for people I respect…
Alto,
I often think, “what do people think psychiatrists did before the age of medications?” Whatever it was, It was how I practiced until I retired 10 years ago. I used some medications, but it was in the single digits of prescriptions per year. Okay, maybe low teens. And it certainly wasn’t time spent dreaming along with Freud and Oedipal Complexes. It was hard work.
Of course that’s not possible now in a free charity clinic where I try to get the meds to rational levels, direct people to the available resources, and do the kind of jake-leg psychotherapy one can do in that setting. I must be getting somewhere because after five years, no non-psychotic patient is on atypicals anymore – and when I came, Seroquel was the rule. I wish I could say that about the people who have fixated on “being bipolar” but aren’t. Their minds are hard to change so I have to accept steady progress rather than success. And thanks to some of your wisdom, people have to taper off of drug A before trying Drug B. Everything is a generic.
But if I lost prescribing privileges tomorrow, there would still be plenty to do. Life is hard…
Mickey-
I do not share your optimism about the rest of medicine. Peter Gotzsche, had many chapters before the one on psychiatry.
Dr. Mickey, even now, psychiatrists who are not inclined towards psychotherapy could do a lot more as doctors besides prescribing psychiatric drugs, such as diagnosing medical conditions and dietary deficiencies that make people think they have psychiatric disorders.
I also see many, many people who firmly believe they are bipolar and have been on drug cocktails for years. This bipolar thing is quite the fad. A friend of mine recently described a pound puppy as “bipolar” because he jumped around in joy when she came home and did the sad eye thing when she was eating.
The sciences in the U.S. are suffering because of a lack of funding and the necessity of getting in bed with a corporation in order to be paid for research. The drugs and uses for them are corrupt with corporate money and KOLs. The drug testings are experimercials and the advertising and marketing is presented as “education” to clinicians.
Also, double blind testing has become synonymous with “scientific” when it’s simply a tactic for limited drug testing for approval. The quality of a scientific pursuit is not in the “answers” but in the questions. Observation has become passé, so all that’s left is measurements and manuals. It’s simple-minded and doesn’t serve the people who are supposed to benefit as well as they would be served if their input and perspectives were valued, considered, and recorded for refererence.
Neurologists had assumed for decades that MS did not cause pain. Obstetricians have thought it best for women to smoke during pregnancy to lower the baby’s weight, in order to make childbirth easier. Doctors use to use x-rays as all-purpose treatments. I had an aunt who died with brain cancer when she was 21. She had been given many very high doses of x-rays to kill ringworms. Doctors have blamed TB on environmental causes for men, and female troubles for women.
But, is there an example in mainstream medicine today in which a person is given a diagnosis based on their reaction to a prescription drug or the personal problems they are having? Does standard medicine tell people their personalities, identities, feelings, and social value can be measured by medical science and determined to be normal or aberrant according to a fifteen question survey?
In studies trying to determine if there is a form of psychosis that is trauma related, psychiatrists are comparing groups of combat veterans they’re considering for the diagnosis, to a group of combat veterans who have been diagnosed with mental illnesses that include psychosis, without questioning the diagnoses of the combat veterans who have been thus labeled. That’s not “objective”. The taxonomy is a series of committee decisions, no more scientific than Baptists voting on whether or not Jesus performed miracles.
The change that is necessary in psychiatry requires that psychiatrists, neurologists, and psychopharmacologists face the fact that their field has become a cargo cult, mimicking science. Medicine has many of these problems, as well, and iatrogenic deaths and injury have become so high that medicine needs to get a grip on itself; but still, medicine is far more grounded in scientific inquiry than psychiatry is.
I agree with you that iatrogenic conditions are under-recognized and often misdiagnosed in psychiatry. Because of the profession’s almost complete reliance on drug treatment, this is far, far more common than in other medical fields.
There is no excuse for this. The adverse effects of psychiatric drugs have been documented since the beginning. Misdiagnosed iatrogenic conditions are entirely due to sloppy clinical practice.
It will be difficult to convince psychiatrists of this, though, because when they misdiagnose, the new diagnosis is invariably another psychiatric disorder. A patient who’s been diagnosed as “bipolar” because of an adverse reaction to an SSRI has no court of appeal. If she seeks a second opinion, it’s likely the second psychiatrist will also mistake the iatrogenesis.
However, the pattern of iatrogenesis and misdiagnosis is crystal clear in this collection of 1,000 case reports from patients http://tinyurl.com/3o4k3j5 You only have to read a few to get the gist. This can happen to anyone.
(See my exegesis of Andrew Solomon, author of The Noonday Demon, and how his “depression” was actually iatrogenic and misdiagnosed https://www.facebook.com/altostrata/posts/619951258058514 )
Iatrogenic conditions and rationalization of them may be far more common in psychiatry than in other medical fields, however in sheer numbers the problem is larger in physical medicine. Patients are told the same things as in psychiatry. My sister, 20 years ago was yelled at to “go ahead and stop taking Prednisone, I don’t care” when she said it wasn’t helping her and the side effects were intolerable. No mention of tapering. Similar situation, I was recently told when I asked specifically about a taper on Metorpolol, that I didn’t have to. However, that was only by a nurse, fortunately my doctor was smarter than that. Another individual I know was told that he had some type of genetic disorder when complaining of extreme muscular pain on a statin. He honestly thought he was dying. Luckily, he missed doses while on vacation and found out that was the med that was causing it. And, afterwards, the doctor went ahead and put him on a different statin, that not surprisingly, caused the same effects.
To me, it is not about which is the worse profession, it is about bigger issues going on in society between patients and health care providers. Issues like blind belief in medical providers followed by viscous feelings of betrayal, expectations of quick fixes, unwillingness to pay for treatments out of pocket, and people en masse not taking ownership of their own healthcare decisions. Note, I am not including people on forced treatment here which brings me to my next point. In mental health, you can add in society’s general lack of interest or funding for anything and the “just not in my backyard” thinking and there is plenty of blame to go around in addition to all the provider issues of incompetence, nativity, laziness, greed, ignorance, and arrogance. Wrap this all up in an insurance industry and government that only serve their own needs and it is a hot mess.
I have no expectations that anyone or even small groups can address these issues fully. Yet, I ask some of you here what are you accomplishing by complaining about how bad psychiatrists are and how they won’t listen to you? So, lets say they are. If we killed all the psychiatrists tomorrow, do you think the issues I mentioned above would go away?
You know the information those of you here provide is invaluable and I have always appreciated it. I have also been reading Mad in America at least once a day for almost a year now and I applaud them for giving voice to people that have no voice. But even over there some are beginning to question the constant complaining and are looking towards the next steps. At some point, the strategy of bashing your head against an immovable wall, pillorying others and tearing down the status quo ceases to be a very effective one in trying to reach the public. It is great that blind faith is destroyed, but what are we supposed to do instead? What choices do people really have? I’d say slim to none. I do know that many of you are working on alternatives, and I guess I really just wish I heard more about them. Maybe then I’d have something more to tell my friends than your antidepressant is all a lie and your psychiatrist is evil.
While ignorance of iatrogenesis occurs in other medical fields, I contend that psychiatry is guilty in the extreme because, for the most part, all psychiatrists do is prescribe drugs. That’s it, the doctor and the prescription pad.
At the very least, they should know the adverse effects of the drugs they hand out all day long. And there are only a few dozen drugs! Sure, many adverse effects have been hidden by pharma, but many are well known and can be easily viewed on http://www.drugs.com/pro/
One cannot count on a psychiatrist, a specialist in the field, to know even what’s been made public by the FDA.