Rarely do you get to see an industry production that has all of these elements rolled into one article, but look no further. This article has all the right moves. It’s a review article in an industry friendly CME journal,
Journal of Clinical Psychiatry. Its got some well known KOLs for guest authors:
Alan Schatzberg former Chairman at Stanford, former
Corcept principle, former APA President, former
Sally Laden client, formerly included on Senator Grassley’s
PHARMA income non-reporter list;
Rakesh Jain, a principle in the CME conglomerate
US Psychiatric and Mental Health Congress; and
Michael Thase,
just-about-everybody’s KOL.
by Alan F. Schatzberg, MD; Pierre Blier, MD, PhD; Larry Culpepper, MD, MPH; Rakesh Jain, MD, MPH; George I. Papakostas, MD; and Michael E. Thase, MD
Journal of Clinical Psychiatry 2014 75[12]:1411–1418.
Six clinicians provide an overview of the serotonergic antidepressant vortioxetine, which was recently approved for the treatment of major depressive disorder in adults. They discuss the pharmacologic profile and receptor-mediated effects of vortioxetine in relation to potential outcomes. Additionally, they summarize the clinical trials, which demonstrate vortioxetine’s efficacy, and discuss findings related to safety and tolerability that have high relevance to patient compliance.
In former times, this kind of industry-driven review article came from "roundtables" – KOL meetings that may or may not have occurred. But it’s different now – teleconferences among the various KOLs.
… highlights from a series of teleconferences held in the spring of 2014. The series was chaired by Alan F. Schatzberg, MD… The faculty were Pierre Blier, MD, PhD…; Michael E. Thase, MD…; George I. Papakostas, MD…; Larry Culpepper, MD, MPH…; and Rakesh Jain, MD, MPH…
The article is obviously ghost-written by a professional medical communications company – apparently paid by the journal.
This evidence-based peer-reviewed Academic Highlights was prepared and independently developed by Healthcare Global Village, Inc., with support from Physicians Postgraduate Press, Inc…
All of the guest authors have extensive industry connections. I’ve just included two from each, the two companies involved with marketing Vortioxetine – 100% onboard.
Financial disclosure: Dr Schatzberg received grant funding and/or honoraria for lectures and/or participation in advisory boards for … Lundbeck/Takeda… Dr Blier received grant funding and/or honoraria for lectures and/or participation in advisory boards for … Lundbeck … Takeda … Dr Culpepper received grant funding and/or honoraria for lectures and/or participation in advisory boards for … H. Lundbeck A/S … Takeda. Dr Jain received grant funding and/or honoraria for lectures and/or participation in advisory boards for … Lundbeck … Takeda. Dr Papakostas received grand funding and/or honoraria for lectures and/or participation in advisory boards for Lundbeck … Takeda. Dr Thase received grant funding and/or honoraria for lectures and/or participation in advisory boards for … H. Lundbeck, A/S … Takeda.
The actual article is odd. For example, it has a discussion of the meanings of NNT and NNH, but neglects to say what Vortioxetine’s NNT actually is. It discusses the changing drug/placebo differences in clinical trials of antidepressants with a borrowed graph, but doesn’t show a graph of any Vortioxetine values. It mentions six positive clinical trials but not the four negative ones.
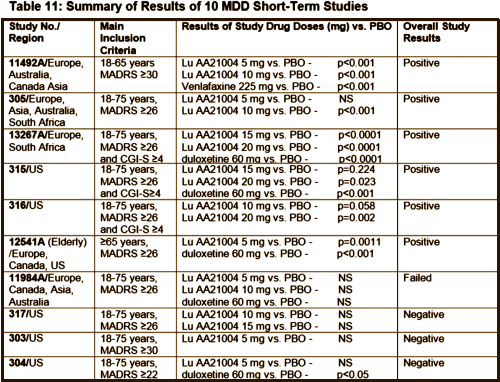
[from the FDA Medical Review]
It even has a discussion of Irving Kirsch’s negative meta-analysis of antidepressant trials and a blurb about how the Affordable Care Act expands mental health benefits. There’s a pitch for
Collaborative Care and encouragement for Primary Care Physicians to prescribe antidepressants. The review ends with:
"Robinson et al in a 2005 study estimated that primary care providers are the sole contacts for more than half of patients with mental illness and that depressive symptoms are present in nearly 70% of patients visiting primary care providers. A patient in a primary care setting, particularly one who has recently acquired mental health coverage through the ACA or Mental Health Parity and Addiction Equity Act, may have one or more coexisting acute or chronic conditions, and to the extent possible, an antidepressant that will not aggravate and might be helpful for these conditions is preferable. A favorable tolerability profile also may improve patient adherence to treatment, leading in turn to not only a reduction in depressive symptoms but a reduced risk of behavioral or social actions that could adversely affect the patient’s overall health and well-being."
So now we have another SSRI on the market called Brintellix® [AKA Vortioxetine], with a review article in the style of so many professionally-written industry-driven articles that have been the flotsam and jetsam of the psychiatric literature for several decades.
At times, when I wear thin with all the anti-psychiatry criticism, the kind that assumes all psychiatrists think the same [stupid] [wrong] things, I’m tempted to get defensive or argue. Then I read an article like this, in a journal like this, by authors like these, and it helps me hold my tongue and turn the other cheek. There’s absolutely nothing here to defend. Fortunately, we don’t see articles like this so much any more, at least in any places that matter. But it’s bad enough that we ever had them in the first place [or that anybody is still writing them].
In the FDA Medical Report on Brintellix®, the reviewer conveniently included a list of all of the FDA approved treatments for Major Depressive Disorder:
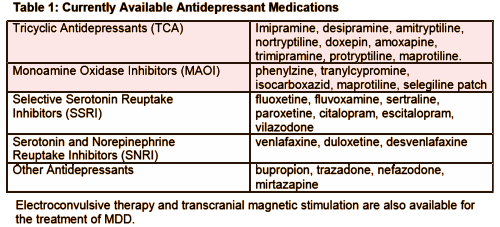
It reminded me of my early days in psychiatry. The drugs available then were the ones in the top two boxes [pink]. In those days, the phrase Major Depressive Disorder would’ve referred to a group of severe conditions I think of as the Melancholic Depressions. I thought of them as diseases, nouns, entities [Depression with a capital "D"]. They aren’t common, but when you see a case, there’s nothing routine about it. The patients are quantitatively profoundly depressed. But there are qualitative features that are striking. It’s as if the affect-generator has been turned to a single frequency – self-hating gloom. Those were the patients we treated with the Tricyclic antidepressants. Sometimes they responded. Sometimes they didn’t. And sometimes they were treated with ECT. People like me who usually spend their days talking to depressed people [depression with a little "d"] are stopped in their tracks when Melancholic Depression is the problem. It’s something else.
When the DSM-III came along in 1980, the phrase Major Depressive Disorder took on a new meaning. If you saw a case of Depression with a capital "D", you could say Major Depressive Disorder with Melancholic features or some such as if there were a continuum between Depression [noun] and depression [symptom] [which isn’t true in my opinion]. Depression had lost its meaning. Any symptomatic depression became known as "MDD" and had graduated – had become a disease [and the most common DSM-III diagnosis at that]. But that’s not the end of the story. In the DSM, there are criteria required to make a diagnosis of "MDD." They’re soft compared to the Melancholic Depressions, but they are at least criteria. So I had a specific reason for posting about this review article, an article I would usually just let pass. But when I read that last paragraph, I started writing:
"… primary care providers are the sole contacts for more than half of patients with mental illness and that depressive symptoms are present in nearly 70% of patients visiting primary care providers. A patient in a primary care setting, particularly one who has recently acquired mental health coverage through the ACA or Mental Health Parity and Addiction Equity Act, may have one or more coexisting acute or chronic conditions, and to the extent possible, an antidepressant that will not aggravate and might be helpful for these conditions is preferable."
According to this article, you don’t even have to meet the watered-down DSM-III criteria for "MDD" to get put on an antidepressant medication anymore. All you have to do is go to your GP for any reason and have some depressive symptoms ["particularly" if you have recently acquired mental health coverage]. Brintellix®, with…
"A favorable tolerability profile also may improve patient adherence to treatment, leading in turn to not only a reduction in depressive symptoms but a reduced risk of behavioral or social actions that could adversely affect the patient’s overall health and well-being."
In an article from the former chairman of psychiatry at Stanford? From a former APA president? That’s the recommendation? Unbelievable!…


There are better sources of information:
http://real-psychiatry.blogspot.com/2014/05/vortioxetine.html
“Using an effect estimation approach the Number Needed To Treat/Harm (NNT/NNH) was 7 (NNT) for response, 11 (NNT) for remission, and 36 (NNH). In the review article the authors describe a total of 10 short term clinical trials and 6 with an active comparator. In 5/6 the active comparator ws duloxetine 60 mg/day and in the other it was venlafaxine extended release 225 mg/day. All 6 of the comparator trials were run against placebo…”
I’m not getting the economics of this from the manufacturer standpoint. Wal-Mart has the older antidepressants at 9 bucks for a three month supply. There is an expensive new sleeper coming out (orexin antagonist) s just as Lunesta is coming off patent.
I just don’t see how these drugs are going to be able to compete unless they are orders of magnitude better than what we have.
BTW, transdermal selegiline is relatively new. That has not taken off in the marketplace despite a novel mechanism of action and the lack of classic MAOI serious side effects and dietary restrictions at low dosage.
Do psychiatrists ever enlist their patients in a treatment method to see if the patient can tell the difference and want to use it? i.e. Do you ever say, “The literature says that there can be some benefit for this drug, but it is not conclusive. It is expensive and has some side effects. Would you be interested in taking it for alternate months with a placebo every other month to see if it is efficacious? I would not tell you which month is the medicine and which the placebo. You would be conducting your own trial. Are you interested?”
My guess is that the drug would have enough side effects that the patient would know the drug from the placebo.
In response to Ferrell Varner’s query, yes – there is some fabulous work by Antonuccio on ADs v placebo – see: Antonuccio DO, Burns DD, Danton WG. Antidepressants: A Triumph of Marketing Over Science? Prevention & Treatment. 2002;5 http://www.antidepressantsfacts.com/2002-07-15-Antonuccio-therapy-vs-med.htm
This is the most incompetent educational (“Academic”) program I have ever laid eyes on. It is a pastiche of segments cobbled together by a clueless medical writer, doubtless using his notes from the teleconferences. Blier’s segment on serotonin receptors is just a collection of factoids that give no meaningful clinical information about vortioxetine; the section on efficacy manages to conceal the response and remission rates with vortioxetine; and as Dr. Mickey pointed out, the section on Number Needed to Treat has gaping holes. Within each segment the main points get lost in the hand waving and in the surrounding, irrelevant wallpaper material.
But, as the article states, the views expressed are those of the named authors – Schatzberg, Blier, Thase, and the rest. Did they do what due diligence required by scrutinizing the final copy? You be the judge! There are no effective linkages between segments – where was maestro Schatzberg, the chairman of the teleconferences? Schatzberg is not so ignorant that he couldn’t see the problems here – was he just lazy and unengaged? As for Thase, he has been burned before by an incompetent CME company because he did not do his due diligence. It looks like it’s back to business as usual in psychiatry KOL land. Does Stanford University have no standards? Does The University of Pennsylvania have no standards?
By the way, a search revealed that the medical communications company Healthcare Global Village, Inc. lists only The Journal of Clinical Psychiatry and The Primary Care Companion for CNS Disorders and Psychiatrist.com as clients. All of these are owned by Physicians Postgraduate Press, Inc. Both companies are based in Memphis. Hmmm.
Sent an e-mail. Hope it didn’t land in a spam filter.
maprotiline listed as both a tricyclic and an MAOI in your table. It is a tetracyclic antidepressant, most accurately listed in the TCA row, but it is not an MAOI!! Someone needs a good proof reader……
Sadly this information will be picked up by a marketing department, repackaged, and then made part of a salesman’s presentation. They will go out and present this to GP’s with the focus on tolerance and helping the unwashed.
This is not directed towards the people on this blog, or even the knuckle dragging business folks like myself, but towards an overworked doctor with just enough knowledge to recognize the names or the organizations involved.
Administrators will see this as another medication to be added to the patient’s regime with the possibility of drug company kick backs, upcoding which can result in higher practice revenue, or even more frequent office visits, again increasing revenue.
Today doctors in practice do not have the time or energy to do their homework on a piece like this, instead they read the abstract or drug reps handout. Critical thinking has been replaced with Google and the instant answer.
With the New Year a day away we need to remember how extraordinary we are as a group, starting with our critical view of all things medical.
Steve Lucas
I’m not sure it even matters if the drug can’t get on the formulary. How does it get on the formulary vs. four dollar a month Prozac?
I’m baffled by the business plans of these new CNS drugs. How are they ever going to recoup R and D? I’ve never seen a patient on EmSam. I’ve seen one on Savella.
Then again I don’t understand the market cap of Twitter and Facebook either.
It’s so quaint to think fundamentals matter. In economics and science.
Ferrelll,
Actually, the issue “the drug would have enough side effects that the patient would know the drug from the placebo” is a major problem in clinical trials themselves.
And thanks for commenting. You and I were lab partners in our Pharmacology Lab in the dawn of time. Who would’ve thought in those days that we would be meeting in a discussion of mis-information in a medical journal? Back then, I guess we thought those articles came down from atop Mount Sinai along with Moses…
Mickey,
Didn’t some of the old school drug trials add a mild anticholinergic as placebo to deal with that issue?
From what I’ve seen from hundreds of patient accounts of their treatments with psychiatric drugs, many prescribers simply like to try something new.
Any antidepressant with a good story, even if manufactured by a marketing ploy, will sell. And if it’s advertised on TV, it will start flying off the shelves tomorrow.
This particular article, a pastiche of sciencey truthiness, exists only to be distributed by drug reps. It will barely be read by prescribers. It’s only a handout to go with samples to get sales going.