by William O. Cooper, S. Todd Callahan, Ayumi Shintani, D. Catherine Fuchs, Richard C. Shelton, Judith A. Dudley, Amy J. Graves, and Wayne A. Ray.Pediatrics 2014 133:1–7.
OBJECTIVES: Recent data showing possible increased risk for suicidal behavior among children and adolescents treated with selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) antidepressants have created significant concern among patients, families, and providers, including concerns about the risk of individual antidepressants. This study was designed to compare the risk for medically treated suicide attempts among new users of sertraline, paroxetine, citalopram, escitalopram, and venlafaxine to risk for new users of fluoxetine.METHODS: A retrospective cohort study included 36842 children aged 6 to 18 years enrolled in Tennessee Medicaid between 1995 and 2006 who were new users of 1 of the antidepressant medications of interest (defined as filling no prescriptions for antidepressants in the preceding 365 days). Medically treated suicide attempts were identified from Medicaid files and vital records and confirmed with medical record review.RESULTS: Four hundred nineteen cohort members had a medically treated suicide attempt with explicit or inferred attempt to die confirmed through medical record review, including 4 who completed suicide. The rate of confirmed suicide attempts for the study drugs ranged from 24.0 per 1000 person-years to 29.1 per 1000 person-years. The adjusted rate of suicide attempts did not differ significantly among current users of SSRI and SNRI antidepressants compared with current users of fluoxetine. Users of multiple antidepressants concomitantly had increased risk for suicide attempt.CONCLUSIONS: In this population-based study of children recently initiating an antidepressant, there was no evidence that risk of suicide attempts differed for commonly prescribed SSRI and SNRI antidepressants.

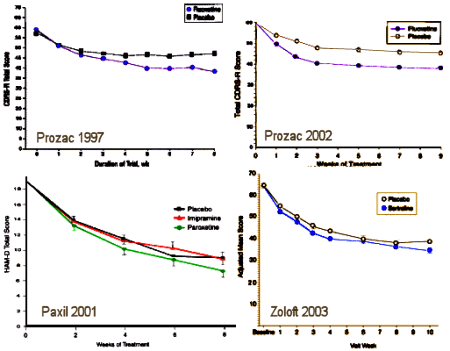
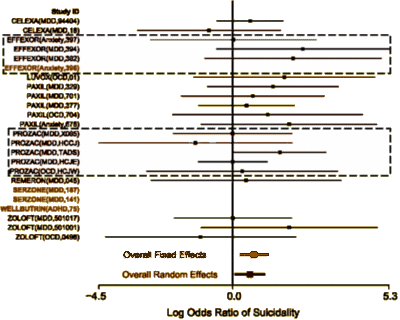
This study is at some level totally absurd, but it points up a glaring piece of craziness in the system we live with. It compares all of the SSRIs to Prozac and finds no difference. Any thought that there would be a difference is a testimony to leftover Eli Lilly advertisement trying to make something out of that artifact of timing. In spite of the FDA’s being unwilling to go along with Lilly, I’ve heard "Prozac is the only SSRI approved for use with adolescents" repeatedly – implying something the FDA never intended. FDA approval is never a treatment recommendation. The efficacy standards for the FDA are low as its primary mission is safety. But the drug companies have used FDA approval as a seal of approval beyond its intent – like some holy writ handed down from the Vatican.
As I understand it, currently fluoxetine is the only SSRI licensed in the UK for the treatment of major depressive disorder (moderate to severe) in children and adolescents. It is assumed that there is evidence to show it carries less risk for this age group.
People regularly sign up on my site SurvivingAntidepressants.org reported severe adverse reactions with only a few doses of an antidepressant. The adverse reactions include unprecedented frightening thoughts, images, and inclinations to self-harm or violence to others.
Quite obviously, there is a population of humans who react very, very badly to serotonergics.
Side note: When a withdrawal syndrome from Prozac became apparent, Eli Lilly convened an “expert panel” led by Alan Schatzberg to study it in 1996. After the filet mignon was served, the “expert panel” declared withdrawal syndrome was a minor nuisance lasting only a few weeks, in a paid-for J Clin Psychiatry supplement http://www.ncbi.nlm.nih.gov/pubmed/9219493 , also see http://www.ncbi.nlm.nih.gov/pubmed/9269249
This whitewash of a conclusion was iterated by a similar “expert panel” in 2004, this time paid for by Wyeth, manufacturer of Effexor, which was making quite the name for itself in difficulty of discontinuation; published in J Clin Psychiatry. 2006. ncbi.nlm.nih.gov/pubmed/16683860
Richard C. Shelton was a member of the second expert panel, and ever after has been a reliable defender of pharmaceutical interests in psychiatry.
Something to consider:
Fluoxetine, paroxetine and to a lesser degree sertraline, are all potent inhibitors of the P40-2D6 enzyme (they are very good at shutting down synthesis) while paradoxically being a substrate of 2D6 (2D6 being their primary pathway for Phase I metabolism). What this means is that at a high enough dose (whatever that is) these drugs shut down their ability to metabolize themselves, sometimes described as “auto” or “suicidal” inhibition (suicidal as in the pharmacological sense) in the literature. Now if you buy into the “wide therapeutic range” nonsense – that tries to say more is better – then you cannot be convinced that ever increasing doses of these drugs will result in ever increasing blood concentrations, which will eventually result in catastrophic adverse events for some people. This says nothing for the known 2D6 genetic polymorphisms – eg. about 10% of Caucasians are considered “poor metabolizers” at 2D6 – and as one knowledgable shrink, on this issue said to me, “if you are a poor metabolizer at 2D6 and prescribed this stuff, you are stuffed” And BTW this is all detailed in the product monograph for Paxil – the large document that most doctors will never read. In any case, my whole point here is the question – why would prozac be any safer
Sorry, important edit to my comment in uppercase square brackets
“then you cannot be convinced that ever increasing doses of these drugs will result in ever increasing [NON-LINEAR] blood concentrations, which will eventually result in catastrophic adverse events for some people.”
And the final sentence Should say “…why would Prozac be considered safer than Paxil?
The danger of inhibition of liver enzymes, particularly 2D6, is well-known regarding antidepressants, but widely ignored in clinical practice.
No matter which psychiatric drug is prescribed, no matter the potential drug-drug interactions, monitoring is poor. Adverse reactions are often overlooked or attributed to pseudo-psychiatric disorders requiring more drugs.
There is nothing good to say about the way psychiatric drugs are currently being prescribed, except that if you owned drug company stock from around 2000 to 2010, you did very well in capital gains.
it seems to me the biggest issue is whether they provide perform as claimed and expected
“The Integrative Management of Treatment-Resistant Depression: A Comprehensive Review and Perspectives
Carvalho A.F. · Berk M. · Hyphantis T.N. · McIntyre R.S.
Psychother Psychosom 2014;83:70-88 (DOI: 10.1159/000357500)
Abstract
Background: Major depressive disorder is a prevalent and disabling illness. Notwithstanding numerous advances in the pharmacological treatment of depression, approximately 70% of patients do not remit after first-line antidepressant treatment. ”
you might think a 70% failure rate would evoke a reaction, a fury, a storm, a debate, a murmur of discontent
However, the drugs decreased libido and sexual response in about 50%, so you can say they are effective in some respects.
jamzo, clinicians take that first-line failure rate to mean they need to batter the brain with more drugs and drug combinations until the patient stops complaining.
ClinicalTrials.gov search results for phase III clinical trials evaluating fluoxetine in children (01/29/2014): how many unpublished studies? how many papers per each clinical trial?
URL: http://t.co/ia91A0C5Gp