One might think from the way this series is going that the next thing would be Trial 0013, the last Clinical Trial submitted to the F.D.A., but in the Clinical Trial world of evidence-based medicine, all is not what it seems to be. And in this story, there’s something missing – Trial 0015. What is Trial 0015? Where is Trial 0015? Why bring up Trial 0015,when Trial 0013 was advertised? So many questions…
The study would come to be called "cursed," but it started out just as Study 15. It was a long-term trial of the antipsychotic drug Seroquel. The common wisdom in psychiatric circles was that newer drugs were far better than older drugs, but Study 15’s results suggested otherwise.
As a result, newly unearthed documents show, Study 15 suffered the same fate as many industry-sponsored trials that yield data drugmakers don’t like: It got buried. It took eight years before a taxpayer-funded study rediscovered what Study 15 had found — and raised serious concerns about an entire new class of expensive drugs. Study 15 was silenced in 1997, the same year Seroquel was approved by the Food and Drug Administration to treat schizophrenia…



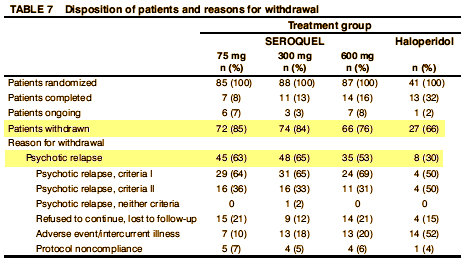
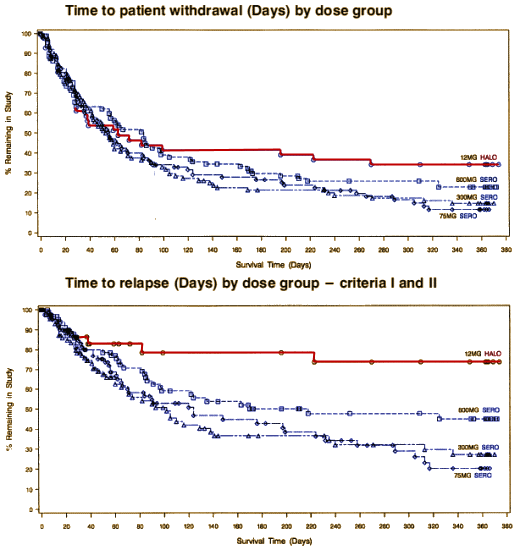
There was no statistically significant dose response among SEROQUEL groups in the time to withdrawal from the trial in the intent to treat population, which was the primary efficacy variable. Pairwise comparison showed no statistically significant differences among any treatment group, including haloperidol, in the time to withdrawal from the trial. There was also no statistically significant dose.response among the SEROQUEL groups in the time to withdrawal from the trial for psychotic relapse. Times to psychotic relapse were generally longer in the haloperidol group. Pairwise comparisons of the time to withdrawal for psychotic relapse revealed statistically significant differences between the haloperidol group and each SEROQUEL group. However, there was an imbalance in the reasons for withdrawal from the trial between the SEROQUEL and haloperidol treatment groups with proportionally more patients in the haloperidol group withdrawing for adverse events. Since proportionally more censoring for relapse occurred in the haloperidol treatment group, any contrasts between the haloperidol and SEROQUEL groups for time to withdrawal for psychotic relapse may not reflect true differences in the relapse distributions among the groups and therefore are non.informative. Results of the analysis of the time to withdrawal in the secondary population showed a similar trend as seen in the intent to treat population. The analysis of prognostic variables indicated that only the interaction between treatment groups and the need for neuroleptic medication to be tapered during Segment A were significantly associated with time to withdrawal.
A little bird tells me that the “Don” mentioned in the “smoke and mirrors” email was indeed a Godfather like figure inside AZ!
How much weight did patients put on in Study 15?
I’m going to look at all the weight gain stuff in a bit. But the answer is going to be “plenty”…
Thank you Mickey. Much appreciated.