 I had a ‘loose end’ from all that looking at the STAR*D study that I was reminded of thinking about "souped up" antidepressants – the CO-MED study that was to follow STAR*D. It was to be a clinical trial where they compared people started on one of a variety of antidepressants to people started on two antidepressants – the list was Wellbutrin, LexaPro, Remeron, and Effexor [a Butyrophenone, an SSRI, a Tetracyclic Antidepressant, and an SNRI respectively]. It’s actually a study I was curious about because I see people in the clinic on combinations of antidepressants all the time [probably started with little more thought than "let’s try this"]. The CO-MED Trial was funded by the NIMH in 2007, started in 2008, and due to end in September 2009. The Principle Investigator, John Rush [also P.I. on STAR*D] left abruptly for Singapore in September 2008 and was replaced as P.I. by his UT colleague Madhukar Trivedi on 12/31/2008.
I had a ‘loose end’ from all that looking at the STAR*D study that I was reminded of thinking about "souped up" antidepressants – the CO-MED study that was to follow STAR*D. It was to be a clinical trial where they compared people started on one of a variety of antidepressants to people started on two antidepressants – the list was Wellbutrin, LexaPro, Remeron, and Effexor [a Butyrophenone, an SSRI, a Tetracyclic Antidepressant, and an SNRI respectively]. It’s actually a study I was curious about because I see people in the clinic on combinations of antidepressants all the time [probably started with little more thought than "let’s try this"]. The CO-MED Trial was funded by the NIMH in 2007, started in 2008, and due to end in September 2009. The Principle Investigator, John Rush [also P.I. on STAR*D] left abruptly for Singapore in September 2008 and was replaced as P.I. by his UT colleague Madhukar Trivedi on 12/31/2008. ![]() The Clinical Trial was recorded as no longer recruiting in February 2009 and as completed in February 2010. The reason for all the details is that it was uncharacteristically very hard to find this study in the NIH RePORTER [usually easy to use] – and once found, the information was often not updated or absent. The other odd thing is that after STAR*D with a gajillion publications, you’d expect we might have heard something from CO-MED. But so far, nada [even though John Rush has been a listed author on 65 articles in PubMed since January 2009]. Here’s what I found:
The Clinical Trial was recorded as no longer recruiting in February 2009 and as completed in February 2010. The reason for all the details is that it was uncharacteristically very hard to find this study in the NIH RePORTER [usually easy to use] – and once found, the information was often not updated or absent. The other odd thing is that after STAR*D with a gajillion publications, you’d expect we might have heard something from CO-MED. But so far, nada [even though John Rush has been a listed author on 65 articles in PubMed since January 2009]. Here’s what I found:
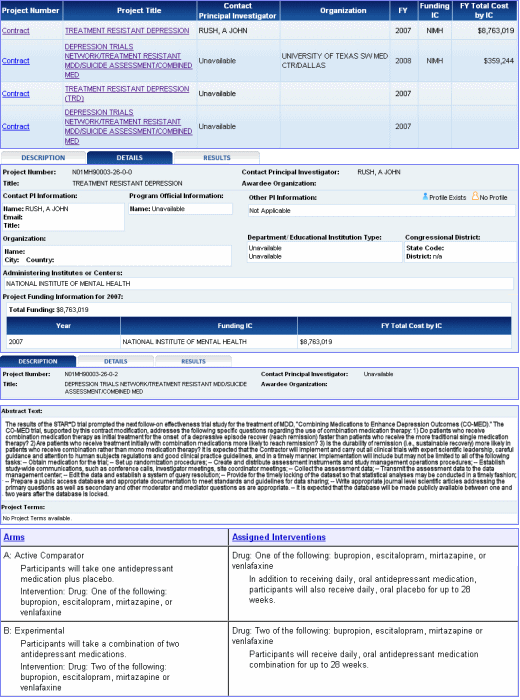
- The results of the STAR*D trial prompted the next follow-on effectiveness trial study for the treatment of MDD, "Combining Medications to Enhance Depression Outcomes (CO-MED)." The CO-MED trial, supported by this contract modification, addresses the following specific questions regarding the use of combination medication therapy: 1) Do patients who receive combination medication therapy as initial treatment for the onset of a depressive episode recover (reach remission) faster than patients who receive the more traditional single medication therapy? 2) Are patients who receive treatment initially with combination medications more likely to reach remission? 3) Is the durability of remission (i.e., sustainable recovery) more likely in patients who receive combination rather than mono medication therapy?
Michael Thase from Star D
http://www.ncbi.nlm.nih.gov/pubmed/16816220?dopt=Abstract
Is still taking money from pharma
http://projects.propublica.org/docdollars/search?term=thase&state%5Bid%5D=
AstraZeneca, Lilly and GSK
I wonder when Americans will demand this practice to stop. Pharma lining the pockets of KOL’s is conflict of interest that is intolerable, and the end result can be something like the scandalous Star*D study. (or the paxil 329 or study 15 seroquel) the list goes on and on.
Instead the news today is Mass is swaying in the wind saying yes to pharma gifts again. I guess the cronies miss their free lunches, pens, notepads and sailboat rides into the sunset ala pharma.
Is everyone tired of this yet? tainted medicine and FDA approved drugs based on cooked/buried/skewed data?
These KOLs are arrogant and should seriously CARE about what the public thinks of them.