Scales like the HAM-D, BDI, MADRS, CDRS, IDS, QIDS, etc are designed to quantify the gamut of depressive symptoms. They’re either administered by a trained rater or self-administered. And they’re used both to certify diagnosis and to follow the progress of treatment in Clinical Trials of MDD [Major Depressive Disorder]. The Q-LES-Q was developed for the second function – following treatment – to measure the subjective experience [Quality of Life] rather than changes in the more objective depressive symptoms. It was collected periodically in the STAR*D Trial:
by IsHak WW, Mirocha J, James D, Tobia G, Vilhauer J, Fakhry H, Pi S, Hanson E, Nashawati R, Peselow ED, Cohen RM.Acta Psychiatrica Scandanavia. 2015 131[1]:51-60.
OBJECTIVE: This study examines the impact of major depressive disorder [MDD] and its treatment on quality of life [QOL].METHOD: From the Sequenced Treatment Alternatives to Relieve Depression [STAR*D] trial, we analyzed complete data of 2280 adult MDD out-patients at entry/exit of each level of antidepressant treatments and after 12 months of entry to follow-up. QOL was measured using the QOL Enjoyment and Satisfaction Questionnaire [Q-LES-Q]. The proportions of patients scoring ‘within-normal’ QOL [within 10% of Q-LES-Q community norms] and those with ‘severely impaired’ QOL [>2 SD below Q-LES-Q community norms] were analyzed.RESULTS: Before treatment, no more than 3% of MDD patients experienced ‘within-normal’ QOL. Following treatment, statistically significant improvements were detected; however, the proportion of patients achieving ‘within-normal’ QOL did not exceed 30%, with >50% of patients experiencing ‘severely impaired’ QOL. Although remitted patients had greater improvements compared with non-remitters, 32-60% continued to experience reduced QOL. 12-month follow-up data revealed that the proportion of patients experiencing ‘within-normal’ QOL show a statistically significant decrease in non-remitters.CONCLUSION: Symptom-focused treatments of MDD may leave a misleading impression that patients have recovered when, in fact, they may be experiencing ongoing QOL deficits. These findings point to the need for investigating specific interventions to ameliorate QOL in MDD.
I wasn’t familiar with the Q-LES-Q. It’s a pretty simple questionnaire, reproduced here FYI:
As the article is short and available on-line, I just clipped out a representative piece illustrating their point that the symptom reduction measured by the QIDS doesn’t directly correlate with an improvement in the Quality of Life. The QOL improvement is much less impressive – consistent with our clinical experience which doesn’t match the often glowing reports in the literature:
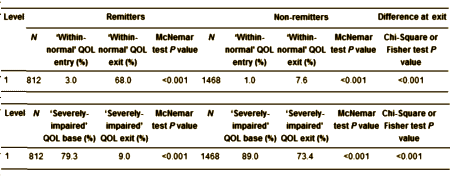
Their punchline:
To conclude, the present analysis highlights the major pitfalls associated with MDD treatments that are purely symptom-focused. Such treatments can give the misleading impression that a patient has recovered, when in fact the patient continues to experience ongoing deficits in QOL. QOL did not improve further after the acute treatment phase even in remitters, and non-remitters showed a statistically significant decline at follow-up after one year. Consequently, clinicians and researchers need to move beyond the mere assessment of symptoms when treating and/or researching MDD, by incorporating QOL measurement, and by investigating specific and personalized interventions to ameliorate QOL.
Like the study in the last post [Depression is not a consistent syndrome…, latter day STAR*D I…], these investigators were able to use the STAR*D dataset available from the NIMH to address a straight-forward question without getting tangled in the problems of the STAR*D study itself. And like in the previous post, one wonders Why didn’t someone do this earlier? It makes intuitive sense that the objective criteria used to define a condition wouldn’t necessarily be the best choice of parameters to follow when assessing subjective improvement. Putting aside for the moment the question of whether Major Depressive Disorder is even a valid diagnostic entity, the antidepressants are, in my mind, symptomatic medications. In the office, at least my office, I don’t go down some checklist of criteria on a follow-up visit after prescribing medication. I let the patient tell me what they think – is it helping or not?
From the day that Max Hamilton introduced the first systematic depression rating scale in 1960 he insisted that it was not a diagnostic instrument. Rather, he intended it to be a measure of severity in patients independently diagnosed as depressed. The HAM-D scale, as it came to be known, also was independent of depressive subtypes (endogenous versus neurotic in those days) and everybody understood that elevated scores could be seen as well in patients with nondepressive diagnoses such as schizophrenia. The reason was that most psychiatric symptoms are diagnostically nonspecific.
When it is used as a severity measure, however, the HAM-D is useful to both researchers and clinicians. During my training I committed the HAM-D to memory, which allowed me to incorporate it seamlessly and unobtrusively into my sessions with depressed patients. In cases where the severity scores came down with treatment but the patients were still complaining that would be a signal to explore further their demoralization or ongoing life stresses and entrapments or their co-morbidities. For our classical patients with recurrent melancholic depression or bipolar I depression, however, the psychometric improvement on the HAM-D was matched by self-report.
My take on these STAR*D data is that the disconnect between the rated psychometric improvement in depression and the quality of life reports may have a lot to do with the kinds of patients who were enrolled in STAR*D. They may have met nominal criteria for the diagnosis of major depression, as discussed in Dr. Mickey’s previous post, on the basis of superficial diagnostic assessments with structured interviews. At the same time they were probably quite different from cases one sees in the clinical referral stream.
One final point: for the most part PhRMA has resisted inclusion of patients’ self-ratings or quality of life measures in clinical trials of antidepressants intended for FDA submission. That’s because those measures seldom agree well with observer ratings of improvement in trials that are managed by CROs. The FDA would do well to reverse this policy.