
 Well, my first seroquel II was premature so I pulled it. Like AstraZeneca itself, I rushed to market before I knew exactly how I wanted to go through the FDA Approval document. This is Version 2.0. What’s new in Version 2.0, you ask? First off, there are a couple of buttons scattered around that pop-up either the FDA 1997 Report on Seroquel itself or my rewrite with essential text and data shown graphically [rather than in imponderable tables]. It’s just kind of useful to have the data at hand so I can talk about it without having to show it over and over. The other difference is that I’m looking at the studies oldest first instead of ‘best’ first.
Well, my first seroquel II was premature so I pulled it. Like AstraZeneca itself, I rushed to market before I knew exactly how I wanted to go through the FDA Approval document. This is Version 2.0. What’s new in Version 2.0, you ask? First off, there are a couple of buttons scattered around that pop-up either the FDA 1997 Report on Seroquel itself or my rewrite with essential text and data shown graphically [rather than in imponderable tables]. It’s just kind of useful to have the data at hand so I can talk about it without having to show it over and over. The other difference is that I’m looking at the studies oldest first instead of ‘best’ first.
The Report I’m looking over is the one prepared for the F.D.A. Approval Panel for Seroquel in February 1997. Zeneca Pharmaceuticals submitted four studies for consideration. The oldest study was Trial 0006, a multicenter six week double blind placebo controlled trial. This study had already been published:
ICI 204,636, an Atypical Antipsychotic: Efficacy and Safety in a Multicenter, Placebo-Controlled Trial in Patients With Schizophrenia by Borison, Richard L. MD, PhD; Arvanitis, Lisa A. MD; Milier, Barbara G. MS. Journal of Clinical Psychopharmacology 1996 Apr;16(2):158-69.
abstract
At the time, Dr. Borison was Chairman of the Department of Psychiatry at the Medical College of Georgia and Dr. Arvanitis and Ms. Miller were on the staff of
Zeneca. This article was accepted in January 1995 and published in April 1996. Two months after this article was published, Dr. Borison abruptly resigned when it was revealed that he was involved in a
massive scam conducting Clinical Trials under the name of the Medical School, but pocketing the money himself [~$10 M]. About the time the F.D.A. was evaluating Seroquel, Borison was being indicted with 172 counts of fraud. He was later convicted and sentenced to 15 years in prison. So I guess this article was his swan-song. In addition, if you have access to the full text, the acknowledgements read like those of "ghost-written" articles [1997 was during the heyday of such shenanigans].
The F.D.A. report describes the trial this way:
"This Phase II trial consisted of one flexible dosing arm of Seroquel (75 mg/day – 750 mg/day) and a placebo arm. The protocol called for 50 patients per arm in order to have 90% power to detect an 8 point difference in change from baseline BPRS between the Seroquel arm and placebo. A total of 109 patients were randomized (Seroquel N=54, Placebo N=55) among 11 centers in the US. However, 3 patients had no post-baseline data leaving 53/group available for analysis. For the 106 patients analyzed, the completion rates were 53% for Seroquel and 42% for placebo."
In the published version, they were more explicit about the dosage:
"Patients randomized to treatment with ICI 204,636 initially received 25 mg three times a day for 1 to 2 days. Thereafter, the dose was titrated upward, using a combination of 25-, 50-, 100-, and 200-mg tablets, until an adequate therapeutic effect was achieved… The maximum daily dose was 750 mg, but daily doses greater than 500 mg were limited to 14 days… Patients randomized to placebo treatment received matching placebo tablets in the same manner as patients treated with ICI 204,636."
I have an agenda in looking at this document. First, I’m really curious how Seroquel got to be such a blockbuster drug approved for so many things, and this seems to be the place to start. Second, I read a lot of those
subpoenaed documents about the deception going on behind the scenes, so I wanted to connect it with the main stage. Finally, I wanted to see how the Clinical Trial scene works, its having occupied such a major role in my profession for the last quarter century of
evidence-based medicine. That said, here’s some of the data from
Trial 0006.
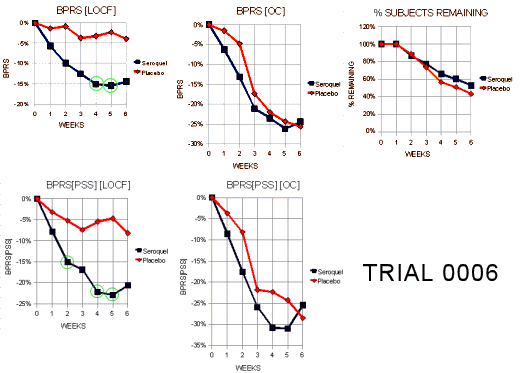
They recorded various outcome rating scales at weekly intervals throughout the study. I graphed the results from the two main scales, the
Brief Psychiatric Rating Scale {BPRS} and its
Positive Symptom Subscale {BPRS[PSS]} – the scale most likely to show changes in psychotic symptoms – as the % fall from the baseline [that’s how such things are usually reported].
We only have the data for the group means in this report, not the raw values, so we can’t directly see the "scatter." We have to rely on the statistical analysis to tell us if differences are meaningful. The significant measurements are circled with green [p<0.05]. Then there are two abbreviations – LOCF [Last Observation Carried Forward] and OC [Observed Cases]. Subjects in Clinical Trials are free agents and they can quit any time they want to. They can quit for reasons that have nothing to do with the study, or because of side effects, or because the treatment doesn’t work, or any of the infinite reasons that motivate mankind – but the point is they do quit with regularity and the investigators have to figure out what to do in their absence. In this study, 42%/53% [Placebo/Seroquel] didn’t complete the whole 6 weeks. A standard method is LOCF – meaning that you use the last observed value for the remainder of the study for the departed subjects. It’s an annoyingly inelegant solution, but it’s hard to figure out another option [If you want to explore the topic, this is a decent discussion]. Fortunately, unlike the statistics, we at least get to see that raw data in the OC graph which only shows the values for the Observed Cases.


On the topic of LOCF, look at the top center graph. It looks like Schizophrenic patients get better on sugar pills. That’s hardly likely. Schizophrenia is bigger than just "placebo effect." What I’d
guess it probably means is that the Schizophrenic subjects that were getting better are the ones that stuck with the study to the end. It’s true for the BPRS[PSS] data as well. In fact, if you click the button that says "GRAPHIC," you can scan down all four studies in this report and see that it’s true for all of them – both the placebo and the treated groups get "more better" over time in the OC graphs compared to the LOCF graphs. I suppose we can
guess that people who are in the study and feeling improvement stay with the program [this is the kind of thing that results is premature balding in Investigators]. Notice also that the only significance is in a few of the weekly measurements, but not at the end of the study. Likewise, the significance is only in the LOCF graph. This is not a particularly interpretable study [in spite of what Borison and Arvanitis said in their published paper]. This alone wouldn’t get you through the F.D.A. [I hope]. Here’s what the reviewers said:
"Tables l0a and l0b display the visit-wise results of the total BPRS for the LOCF and observed cases analyses, respectively. The borderline statistical result for the LOCF is likely due to the less than anticipated treatment difference at 6 weeks (-8.1 vs -2.1). Note that there was no difference at all for the completers. Thus, Figure 5 indicates that the entire treatment difference in the LOCF analysis is due to dropouts in the first 4 weeks of the trial."
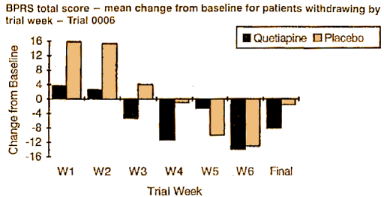
The reviewers had the raw numbers. They compiled the "last observations" that were "carried forward." What they could see was that those early drop-outs in the placebo groups who were getting worse added a skew to the graphs that pushed it into the realm of guessing instead of science. That’s not Zeneca‘s fault. That’s the real world. In ancient times when all of these patients would have been housed in a State Mental Hospital, they could’ve gotten data on 100% of the participants. It would’ve been like the animal room in the Psychology Professor’s laboratory. Then there would only be one graph – Observed Cases – with no "corrections" required. In that case, the heterogeneity of the Schizophrenic patients that probably explains their results and throws us into the guessing game would have been amenable to scientific analysis. Such things are usually managed by having large subject cohorts in lots of different sites [which costs a bundle]. They project [guess at] such things mathematically:
"Power calculations were based on the ability to detect a minimum difference of 8 units between mean changes from baseline in BPRS total scores. A variance estimator from a previous study (mean square error of 123.9; data on file, ZENECA Pharmaceuticals) was used to determine that 50 patients per treatment group would allow detection of this difference with a power of at least 0.90 and a two-sided alpha = 0.05 level test."
It looks as though they missed this time. Trial 0006 would not get the job done with a reviewer who had all the facts [the raw data]. The "drop-out" variable and the heterogenity of their Schizophrenic subjects carried the day. But all the information wasn’t available to someone reading the paper that Drs. Borison and Arvanitis published. Embedded in the text they mention the drop-outs:
Twenty-six patients treated with ICI 204,636 and 33 placebo-treated patients withdrew before completing 6 weeks of treatment for reasons shown in Table 1. The majority of withdrawals [16 in the ICI 204,636 group; 27 in the placebo group] were attributed to treatment failure.
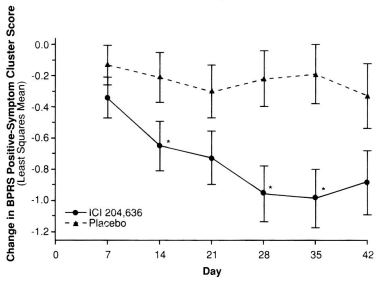
But they don’t show the impact of the drop-outs on the data. They simply show us their version of the LOCF graph:
And they conclude:
In this placebo-controlled, multicenter trial, ICI 204,636 was effective in the treatment of the positive and negative symptoms of schizophrenia. Significant differences (p less or equal to 0.05) between treatment groups were identified for the primary efficacy variables, the BPRS total score and CGI Severity of Illness item score, at most times throughout the trial, with marginal significance achieved at endpoint (p = 0.07 for both)…
Maybe. Maybe not. With Trial 0006, we can’t say either way that Seroquel is what what Zeneca wants it to be. And from their published results, we wouldn’t know that it’s a guessing game at this point. But as long as they’ve thrown us into the world of guessing, I’m going to make a guess too. My guess is that the published study paid for by Zeneca and co-authored by two Zeneca employees and America’s legendary Academic-Psychiatrist-for-hire, Dr. Borison [at the height of his Madoff-like career], was ghostwritten:
This trial was supported by a grant from ZENECA Pharmaceuticals, Wilmington, DE. SEROQUEL is a trademark, the property of ZENECA Limited. We thank Suzanne Bristow-Marcalus, RPh, ELS, for medical writing support; Jean Fennimore for trial organization and management support; Jacqueline Fiore and Gary Cooper for trial monitoring support; and Kathleen Jelliffe, Denise Redkar-Brown, Joy Russo, and Thais Womack for data management support.
But I can’t prove that anymore than Trial 0006 proves Seroquel’s efficacy. Three+ studies to go…

 Well, my first seroquel II was premature so I pulled it. Like AstraZeneca itself, I rushed to market before I knew exactly how I wanted to go through the FDA Approval document. This is Version 2.0. What’s new in Version 2.0, you ask? First off, there are a couple of buttons scattered around that pop-up either the FDA 1997 Report on Seroquel itself or my rewrite with essential text and data shown graphically [rather than in imponderable tables]. It’s just kind of useful to have the data at hand so I can talk about it without having to show it over and over. The other difference is that I’m looking at the studies oldest first instead of ‘best’ first.
Well, my first seroquel II was premature so I pulled it. Like AstraZeneca itself, I rushed to market before I knew exactly how I wanted to go through the FDA Approval document. This is Version 2.0. What’s new in Version 2.0, you ask? First off, there are a couple of buttons scattered around that pop-up either the FDA 1997 Report on Seroquel itself or my rewrite with essential text and data shown graphically [rather than in imponderable tables]. It’s just kind of useful to have the data at hand so I can talk about it without having to show it over and over. The other difference is that I’m looking at the studies oldest first instead of ‘best’ first.

 On the topic of LOCF, look at the top center graph. It looks like Schizophrenic patients get better on sugar pills. That’s hardly likely. Schizophrenia is bigger than just "placebo effect." What I’d guess it probably means is that the Schizophrenic subjects that were getting better are the ones that stuck with the study to the end. It’s true for the BPRS[PSS] data as well. In fact, if you click the button that says "GRAPHIC," you can scan down all four studies in this report and see that it’s true for all of them – both the placebo and the treated groups get "more better" over time in the OC graphs compared to the LOCF graphs. I suppose we can guess that people who are in the study and feeling improvement stay with the program [this is the kind of thing that results is premature balding in Investigators]. Notice also that the only significance is in a few of the weekly measurements, but not at the end of the study. Likewise, the significance is only in the LOCF graph. This is not a particularly interpretable study [in spite of what Borison and Arvanitis said in their published paper]. This alone wouldn’t get you through the F.D.A. [I hope]. Here’s what the reviewers said:
On the topic of LOCF, look at the top center graph. It looks like Schizophrenic patients get better on sugar pills. That’s hardly likely. Schizophrenia is bigger than just "placebo effect." What I’d guess it probably means is that the Schizophrenic subjects that were getting better are the ones that stuck with the study to the end. It’s true for the BPRS[PSS] data as well. In fact, if you click the button that says "GRAPHIC," you can scan down all four studies in this report and see that it’s true for all of them – both the placebo and the treated groups get "more better" over time in the OC graphs compared to the LOCF graphs. I suppose we can guess that people who are in the study and feeling improvement stay with the program [this is the kind of thing that results is premature balding in Investigators]. Notice also that the only significance is in a few of the weekly measurements, but not at the end of the study. Likewise, the significance is only in the LOCF graph. This is not a particularly interpretable study [in spite of what Borison and Arvanitis said in their published paper]. This alone wouldn’t get you through the F.D.A. [I hope]. Here’s what the reviewers said:

The part 2 rewrite is much better reading; more steak and potatoes & easier to understand.
Thank you for taking the time to break this all down for us. The public needs to know how AstraZeneca & other pharmaceutical giants create these blockbuster drugs.
This series could really be called “unveiling the Emperors new clothes”. .
Thanks for breaking it down, and for your analysis—I can’t help but feeling more than a little sick from what I am learning. The betrayal is immense and overwhelming.
Wish I had never been given this drug!