THE SUICIDE STATISTICS
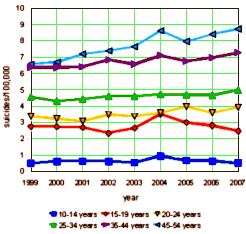 The suicide numbers came from the CDC Compressed Mortality, 1999-2007 Results database. The image on the left are the suicide rates [per 100,000] for females at various age groups over the period available [the absolute number graph looks essentially the same]. Parenthetically, when I look at the graph, the most striking thing is the steady increase in the suicide rate in women in the two groups over 35 y/o. There is something of a blip in 2004 in both the over 35 y/o and under 20 y/o groups. The National Bureau of Economic Research study we’re reviewing focuses attention on that blip in 2004 and is going to compare the 10-19 y/o group [red+blue] to the 20-24 y/o group [yellow] and ignore the others.
The suicide numbers came from the CDC Compressed Mortality, 1999-2007 Results database. The image on the left are the suicide rates [per 100,000] for females at various age groups over the period available [the absolute number graph looks essentially the same]. Parenthetically, when I look at the graph, the most striking thing is the steady increase in the suicide rate in women in the two groups over 35 y/o. There is something of a blip in 2004 in both the over 35 y/o and under 20 y/o groups. The National Bureau of Economic Research study we’re reviewing focuses attention on that blip in 2004 and is going to compare the 10-19 y/o group [red+blue] to the 20-24 y/o group [yellow] and ignore the others.
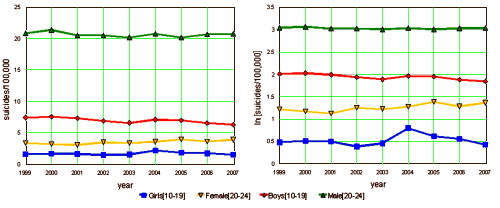
To better understand how the FDA activity around pediatric antidepressant use may have affected symptoms in the population of adolescents with depression, Figure 4 presents information regarding suicides in the United States over the period from 1999 to 2007. These data are drawn from the Centers for Disease Control and Prevention’s publicly available information on deaths by cause and selected demographic characteristics. Because of dramatically different levels of suicides by age and gender [males commit suicide more often, and between ages 10 and 24, suicide rates rise with age], the figure presents the natural log of suicide deaths per 100,000 population, separately for two age groups [10-19 and 20-24], and by sex. Because the 2004 FDA warnings applied to pediatric antidepressant use, they did not directly affect 20-24 year olds. Youth suicides had been flat or declining among 10-19 year olds in the years preceding the warnings, but in 2004, 10-19 year old girls experienced a sharp increase in suicides, of over 30 percent. There was no abrupt rise among older ages [20-24] as expected, given that these early warnings focused on pediatric populations.
THE MEDIA COVERAGE
News coverage of FDA warnings on pediatric antidepressant use and suicidality
by Barry CL and Busch SH.
Pediatrics. 2010 125[1]:88-95.
[full text online]
OBJECTIVE: In 2004, after an 18-month investigation, the Food and Drug Administration [FDA] directed pharmaceutical manufacturers to add a black box warning to antidepressants regarding increased risk of suicidality in children. Our objective was to evaluate the quality and content of and overall impression conveyed in news coverage of this issue.
METHODS: We collected all news stories on pediatric antidepressant use and suicidality published in a convenience sample of 10 of the highest-circulation print newspapers in the United States, the 3 major television networks, and a major cable news network in 2003 and 2004 [N = 167]. Two researchers coded news articles by using a 9-item instrument.
RESULTS: The quality of news reporting on key health messages included in FDA warnings was mixed. The overwhelming majority of news stories correctly described a risk of suicidality associated with pediatric antidepressant use, as opposed to suicide itself. However, other key health messages highlighted in FDA warnings often were absent from news coverage. News stories were more likely to include anecdotes of children harmed versus children helped by antidepressants, whereas expert sources quoted were more likely to emphasize the benefits of antidepressants over their risks. However, the majority of news stories conveyed neither the overall impression that the risks of pediatric antidepressant use outweighed the benefits nor the impression that the benefits outweighed the risks, and coverage became increasingly neutral over time.
CONCLUSIONS: Inclusion of key health messages in FDA safety warnings was not sufficient to ensure their communication to the public through the lay press.
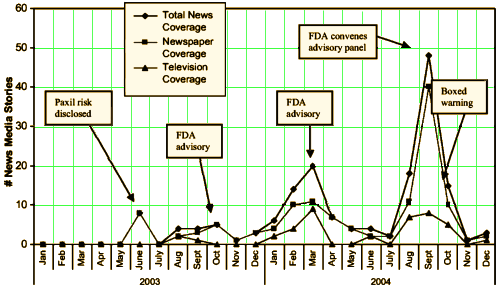
[reformatted to fit the page]
Because depression is an undertreated disease with the potential for long-term negative consequences, the sharp decreases in pediatric antidepressant use that occurred in the aftermath of the FDA risk disclosure are troubling, to the extent that they represent an increase in unmet need. Clinical trial data indicated that one antidepressant, fluoxetine, was clinically effective in treating children with major depressive disorder. The efficacy of other antidepressants in treating children has not been proved. In the 1990s, increased antidepressant use after the introduction of SSRIs was hailed by many as an important advancement in public health. Steady decreases in youth suicide rates suggested to some that antidepressants were saving lives. This trend reversed unexpectedly with the upswing in national youth suicide rates in 2004 and 2005, leading to the speculation that less antidepressant use after FDA safety warnings might have increased rather than decreased youth suicide rates, although other explanations also are possible [eg, changes in youth access to firearms, the prevalence of alcohol use, and Internet-based social networks].
THE PRESCRIPTION RATES FOR ADOLESCENTS
So why all the focus on the media reports? The Black Box warnings weren’t mandated until October 2004, so they couldn’t have had that much effect on the 2004 suicide statistics. And since this Report is determined to show that the FDA warnings essentially caused a suicide increase in adolescents in 2004, they would have to show that all the media noise caused a drop in prescriptions for adolescents before they were mandated.
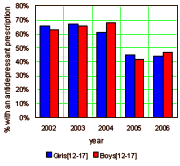 In fact, one of the slides in the NBER paper [reproduced on the right using their numbers] would suggest that prescribing didn’t fall until 2005, negating their argument altogether. But that was not the only shot at trying to quantitate a fall in antidepressant prescribing in adolescents that lasted throughout 2004 which might support their hypothesis. One such study is one I’ve already reviewed [the apogee…]. If you review the graph, the reported prescription rate does fall early in the year in that cohort. Now, have a look at what the authors have to say. I’ve included their Appendix with links to the particular studies they chose to focus on.
In fact, one of the slides in the NBER paper [reproduced on the right using their numbers] would suggest that prescribing didn’t fall until 2005, negating their argument altogether. But that was not the only shot at trying to quantitate a fall in antidepressant prescribing in adolescents that lasted throughout 2004 which might support their hypothesis. One such study is one I’ve already reviewed [the apogee…]. If you review the graph, the reported prescription rate does fall early in the year in that cohort. Now, have a look at what the authors have to say. I’ve included their Appendix with links to the particular studies they chose to focus on.
…The most often cited of these studies are summarized briefly in Appendix Table I. Studies that focus on monthly antidepressant sales demonstrate an initial abrupt decline in prescribing of antidepressants in pediatric populations beginning in January-March of 2004, the time of public hearings on the safety of pediatric antidepressant use [Benji T Kurian, Wayne A. Ray, Patrick G. Arbogast, D. Catherine Fuchs, Judith A. Dudley and William O. Cooper, 2007, Charles B. Nemeroff, Amir Kalali, Martin B. Keller, Dennis S. Charney, Susan E. Lenderts, Elisa F. Cascade, Hugo Stephenson and Alan F. Schatzberg, 2007]. One study of all-payer data representing about half of all prescription activity in the U.S., shows the precipitous drop in the number of prescriptions for children under age 18 that occurred in January-June 2004 coincident with intense media coverage of FDA’s hearings and its second advisory on safety risks of pediatric antidepressant use [Charles B. Nemeroff, Amir Kalali, Martin B. Keller, Dennis S. Charney, Susan E. Lenderts, Elisa F. Cascade, Hugo Stephenson and Alan F. Schatzberg, 2007].
| Appendix Table I. Peer-reviewed studies of declines in pediatric antidepressant use in the United States |
|||
| Citation | Population/Data | Results | |
|
|
|||
| * | Nemeroff et al., 2007 | Verispan data; represents ~ ½ of retail prescriptions in US [ages 0-17] |
5% decline in pediatric antidepressant volume in Q1 2004; Additional 11% decline in Q2 2004; No further change through March 2005 |
| * | Gibbons et al., 2007 | IMS pharmacy data; aggregated to national level [ages 0-14] |
22% decline in youth antidepressant volume from 2003 to 2005; 30% decline in new youth antidepressant prescription volume from 2003 to 2005 |
| * | Kurian et al., 2007 | Tennessee Medicaid pharmacy data; [ages 2-17] |
33% decline in new antidepressant use [i.e. individual fills any antidepressant prescription in month] |
| Busch et al., 2010 | Medical Expenditure Panel Survey; nationally representative survey data [ages 5-21] |
26% decline in twelve-month prevalence rates of antidepressant use from 2002/3 to 2005/6 [i.e., individual fills any antidepressant prescription in year] | |
| * | Libby et al., 2009 | PharMetrics Patient-Centric Database; eighty-five managed care plans [ages 5-18] |
After accounting for prescribing trends, 58 % decline in antidepressant use among individuals newly diagnosed with depression [within 30 days post diagnosis.] |
| * | Busch et al., 2010b | Marketscan data; privately insured [ages 5-17] |
19% decline in antidepressant use among individuals newly diagnosed with depression [within 30 days post diagnosis.] |
| Valluri et al, 2010 | i3 Innovus database; commercial insurance enrollees [ages 2-17] |
17% decline in antidepressant use among individuals newly diagnosed with depression [within 180 days post diagnosis.] | |
Could there be anything more to say on this issue? Plenty more, but I’m only going to add one more piece, the comments from a Lawyer in the firm that has handled many of the big lawsuits against the Pharmaceutical Manufacturers:
NBER’s Contentions Regarding the Level of Prescriptions and Suicides Are Factually Flawed
Baum Hedlund
by Bijan Esfandiari
September 30, 2011Moving past the efficacy issue, NBER’s contention that youth suicides have increased following the advent of the black-box warning is also factually flawed. The Reason article cites NBER’s fallacious working paper for the proposition that “Youth suicides had been flat or declining among 10-19 year olds in the years preceding the warnings, but in 2004, 10-19 year old girls experienced a sharp increase in suicides, of over 30 percent.” This is simply not supported by the data, which, as outlined below, reveal that suicide rates were not only rising prior to issuance of the black box warnings, but actually went down following issuance of the black box warnings.
In 2004, the year suicides increased, “there was no significant drop in SSRI prescribing." [The Black Box Warning: Decreased Prescriptions and Increased Youth Suicide? by JON JUREIDINI]. As Jureidini pointed out, Gibbons “incorrectly analyzed the relationship” between prescription rates and suicide rates among children and, when carefully examined, Gibbons’ suggestion of a clear causal association from the data analyzed “show no such association.” Id. Like Gibbons, the NBER working paper is making erroneous “[a]larmist predictions regarding the consequences of decreased prescribing.” Id.
Gibbons, himself, has since acknowledged that his data did not support a causal link: “We really need to see the 2005 numbers on suicide to see what happened." [Experts Question Study on Youth Suicide Rates] (The alleged culprit of the NBER’s “list of terribles,” i.e., the black box warning, was implemented in early 2005, and antidepressant prescriptions did decrease that year.) The FDA’s Dr. Thomas Laughren confirmed at a 2008 hearing that antidepressant prescribing did not change until 2005 and the 2005 suicide numbers “are down from where they were in 2004."
Moreover, one of the studies cited by the working paper authors by Gibbons et al. was so scientifically illegitimate that it was described by the British Medical Journal as “astonishing,” “misleading” and "reckless." [Dutch academics criticise suicide claims made in American psychiatry journal]. Further, one of the Gibbons study authors, Ron Herings admitted the study’s findings are “not right” and that it “doesn’t follow from the data, it is not true and serves just to scare people. It is hard to admit this, as I am one of the authors of the article and I attached my name to it …" [Interview with Ron Herings, Argos, VPRO/VARA, Radio 1 (December 7, 2007)].
Finally, NBER’s reliance upon ecological studies to support their arguments is methodologically flawed. Authors of ecological “studies” universally agree you cannot conclude that increasing or decreasing suicides (or any other malady) is the result of a lack of antidepressant consumption based on these types of analyses.
 This whole story is truly remarkable – particularly in light of the simple set of suicide statistics on the right that underpins the whole discussion. The hypothesis is that the decision to append a black box warning had all kinds of negative repercussions – including an increase in teen suicides. The only evidence was that 2004 increase in the red line. Since the warning came at the end of that year, they propose that the media reporting lead to a pre-emptive decrease in prescribing, and various records were produced to show this was indeed the case. But when you look at the graph, the suicide rate for teenaged girls is now falling as opposed to the over 35 y/o groups which definitely seem to be on the rise [groups still taking antidepressants].
This whole story is truly remarkable – particularly in light of the simple set of suicide statistics on the right that underpins the whole discussion. The hypothesis is that the decision to append a black box warning had all kinds of negative repercussions – including an increase in teen suicides. The only evidence was that 2004 increase in the red line. Since the warning came at the end of that year, they propose that the media reporting lead to a pre-emptive decrease in prescribing, and various records were produced to show this was indeed the case. But when you look at the graph, the suicide rate for teenaged girls is now falling as opposed to the over 35 y/o groups which definitely seem to be on the rise [groups still taking antidepressants].I don’t know why the suicide rate was higher in 2004 for teens [or for that matter in women over 35 y/o]. But it seems silly to continue to claim that depressed kids are killing themselves by being deprived of SSRIs – for the obvious reason that they’re not. It’s not happening. The SSRIs don’t seem to be doing that much for the over 35 y/o set either. And if they want to blame that 2004 blip on something, how about withdrawal symptoms from stopping them too quickly? – something else the drug manufacturers denied along the way [see, I can make up stuff too]. This continued assault on the FDA warnings is yet another example of the direct intrusion of the drug industry into the psychiatric literature – a conclusion in search of an argument. Simply looking at the suicide statistics disproves their premise without any of the window dressing.
They suggest that the unintended consequences of policies designed to protect consumer safety extend well beyond the clinical considerations that typically contribute to decisions about the costs and benefits of regulatory actions. The FDA’s decision to issue the warning on antidepressants rested on evidence of suicidal thoughts and actions, but not suicide itself. Indeed, the FDA panel of experts convened to make the decision about a black box warning was divided regarding the relative costs and benefits of issuing the warning for public health. The “costs” of issuing the warning considered were clinical in nature, with potential suicide among depressed patients being the most extreme of these. However, the discussion did not consider the broader non-clinical costs [and benefits] of treating depression with antidepressants. Second, our findings also confirm research in very different settings suggesting that depression interferes with academic outcomes, especially for girls.The third and most significant innovation of this empirical work is that it suggests that treatment of depression can mitigate adverse effects of depression on both cognitive and non cognitive aspects of human capital among adolescents. Families, schools, and clinicians devote significant effort to help students with behavioral health issues because they interfere with academic outcomes. These findings suggest that treatment of depression can improve both cognitive outcomes such as grades, as well as delinquent behaviors and the initiation of substance use, all of which are important aspects of human capital. Our paper focuses on outcomes that occurred within 1 to 12 months of being surveyed, and thus they should be viewed as relatively short-run outcomes. Given controversy over the risks and benefits of antidepressants in the long run, our findings do not suggest we should abandon other forms of treating depression, but rather as evidence that depression affects economic outcomes, and thus successful treatments of depression yields potential benefits that reach far beyond the clinical outcomes typically measured.
The job of the FDA is to certify the safety and efficacy of drugs available for prescription, and to inform physicians about the adverse effects of the drugs they use. To suggest that the FDA withhold a warning of a potentially dangerous adverse effect of a medication based on the considerations in these concluding remarks betrays either a massive misunderstanding of the practice of medicine or a wish to totally change our basic system. If they want to get doctors prescribing SSRIs to adolescents in spite of the possible adverse reactions, talk to the doctors, not the FDA. I know why the compromised KOLs fought this decision and keep hammering at it. They were colluding with the pharmaceutical companies to increase drug sales. I would assume that the National Bureau of Economic Research has a vested interest in that their leader is on the Board of Eli Lilly. But this article and some that preceded it are from health policy academics at Yale. I can only conclude that the drug companies have found a way to get to them too. They should know better.
According to the CDC, ELEVEN PERCENT!! of the U.S. adult population, more women than men, are now taking antidepressants, mostly SSRIs. Over 60% of those taking such drugs have been taking them for over two years, but fewer than 1/3 of those taking a single antidepressant have seen a mental health professional in the past year. On the other hand, only 1/3 of those with symptoms of severe depression — the group in which the drugs might actually be more effective than a placebo, are taking such drugs.
See:
http://www.cdc.gov/nchs/data/databriefs/db76.htm
Antidepressants now seem to getting handed out “for what ails you” as if they were M&Ms.
According to an article in AMA Medical News, “Practical approaches to mental health (AAFP Scientific Assembly)”, “Family physicians are urged to screen their patients for mental illness AND TREAT THOSE WHOSE CONDITIONS ARE NOT SEVERE.” And since Family Physicians don’t have much time, or much training in doing psychotherapy, it appears that they will be prescribing antidepressants for those patients with non-severe depression — just the group in whom antidepressants are least likely to be more effective than placebos. [emphasis added]
See:
http://www.ama-assn.org/amednews/2011/10/17/hlsa1017.htm
So I guess that 11% marketing success is just a beginning.
And now, we hear that there is now some evidence which MIGHT link widespread antidepressant use by pregnant women with the mysterious sharp rise in autistic spectrum disorders in “first world” countries:
See:
http://psychcentral.com/news/2011/10/26/rat-study-ties-antidepressants-to-autism-like-brain-abnormalities/30761.html
See:
http://www.foxnews.com/health/2011/10/24/prenatal-exposure-to-antidepressants-makes-rats-act-autistic/
From one of the above cited articles:
“The incidence of pregnant women taking SSRIs has grown from about .5 percent in 1985 when the first one came on the market to nearly 10 percent today, Paul said.
“The rates of incidence of Autistic Spectrum Disorders have roughly doubled every three to five years over the past decade with a current incidence of 1 in every 91 births, say the researchers. By contrast, in 1996, the rate of incidence was less than 1 in 1,000 births and by 2007 it reached about 1 in 200.”
http://psychcentral.com/news/2011/10/26/rat-study-ties-antidepressants-to-autism-like-brain-abnormalities/30761.html
I think the most creative part of the data manipulation is the completely ludicrous suggestion that a black box warning which appeared in October of 2004 could have anything to do with suicide rates in 2004. Ridiculous. And I kind of like it when you make up stuff too.