
[This is tedious, but somebody has to do it]. Back in March, Speilmans et al published a meta-analysis of the studies Augmenting Antidepressants with Atypical Antipsychotics in cases of inadequate response [Adjunctive Atypical Antipsychotic Treatment for Major Depressive Disorder: A Meta-Analysis of Depression, Quality of Life, and Safety Outcomes]. I spent some time on that meta-analysis [and…, the usual suspects…] because the whole idea makes so little sense to me. Why should an Antipsychotic have much to do with depression? What does Augmentation mean anyway? Having looked recently at the studies using Seroquel XR® as monotherapy in Major Depressive Disorder and concluding that I thought whatever effect it had on depression was due to improved sleep [seroquel: good to the last drop…, rest my case…, another thing…] I made a note to myself to look at those augmentation studies again with the same question in mind.
by Bauer M, El-Khalili N, Datto C, Szamosi J, and Eriksson HJournal of Affective Disorders. 2010 127[1-3]:19-30.
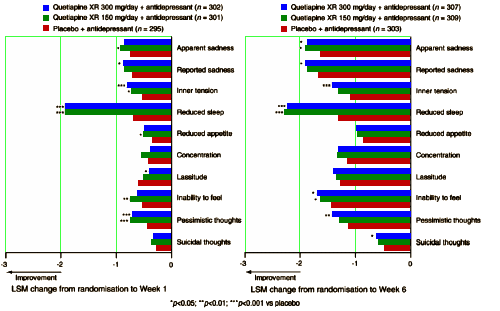
BACKGROUND: Two positive studies evaluated adjunctive extended release quetiapine fumarate (quetiapine XR) in patients with major depressive disorder (MDD) showing inadequate response to antidepressant treatment. This preplanned, pooled analysis provides an opportunity for subgroup analyses investigating the influence of demographic and disease-related factors on observed responses. Additional post hoc analyses examined the efficacy of quetiapine XR against specific depressive symptoms including sleep.METHODS: Data were analysed from two 6-week, multicentre, double-blind, randomised, placebo-controlled studies, prospectively designed to be pooled. Patients received once-daily quetiapineXR 150mg/day (n=309), 300mg/day (n=307) or placebo (n=303) adjunctive to ongoing antidepressant therapy. The primary endpoint was change from randomisation to Week 6 in MADRS total score. Other assessments included MADRS response (≥50% decrease in total score) and remission (total score≤8), change from randomisation in HAM-D, HAM-A, PSQI global and CGI-S scores.RESULTS: Quetiapine XR (150 and 300mg/day) reduced MADRS total scores vs placebo at every assessment including Week 6 (-14.5, -14.8, -12.0; p<0.001 each dose) and Week 1 (-7.8,-7.3,-5.1; p<0.001 each dose). For quetiapineXR 150 and 300mg/day and placebo, respectively at Week 6: MADRS response 53.7% (p=0.063), 58.3% (p<0.01) and 46.2%; MADRS remission 35.6% (p<0.01), 36.5% (p<0.001) and 24.1%. QuetiapineXR 150 and 300mg/day significantly improved HAM-D, HAM-A, PSQI and CGI-S scores at Week 6 vs placebo. Quetiapine XR demonstrated broad efficacy, independent of factors including concomitant antidepressant.CONCLUSIONS: Adjunctive quetiapine XR is effective in patients with MDD and an inadequate response to antidepressant therapy, with improvement in depressive symptoms seen as early as Week 1.
[recolored and reformatted for clarity]
[recolored and reformatted for clarity]
…The early improvement seen with quetiapine XR in the current study may appear to be in part due to a beneficial effect on sleep since among the individual MADRS items, the greatest magnitude of improvement at Weeks 1 and 6 was observed in Item 4 (reduced sleep). Quetiapine XR was also associated with significant improvement in PSQI global score, MADRS Item 4 (reduced sleep) and HAM-D sleep disturbance factor scores at Week 6. However, it is of interest to note that the results from the post hoc analysis to determine whether treatment effects on sleep were independent of effects on the primary endpoint relationship between sleep and efficacy showed a significant overall improvement in MADRS total score at Week 6. This demonstrates the broad effects of quetiapine XR across a range of symptoms of depression which occur independently of its effect on sleep. Beyond improvements in sleep quetiapine XR also improved other MADRS items at Week 1.
Significant improvements in MADRS total score were seen in patients, irrespective of whether or not they experienced an AE potentially related to somnolence, suggesting that any concomitant sedative effects do not interfere with the antidepressant effect of quetiapine XR. This observation further supports our finding that quetiapine XR antidepressant properties are independent of treatment effects on the core symptom of reduced sleep in patients with MDD.
 The highlighted parts are intended to convince us that Seroquel XR® has antidepressant properties independent of its sedative properties. Needless to say, I was not so moved [in fact, even rereading it to see if I was missing something made me sleepy]. AstraZenecal has let the cat out of the bag with these studies because when I look at this data, I see SLEEPING PILL, and that’s about all I see…
The highlighted parts are intended to convince us that Seroquel XR® has antidepressant properties independent of its sedative properties. Needless to say, I was not so moved [in fact, even rereading it to see if I was missing something made me sleepy]. AstraZenecal has let the cat out of the bag with these studies because when I look at this data, I see SLEEPING PILL, and that’s about all I see… 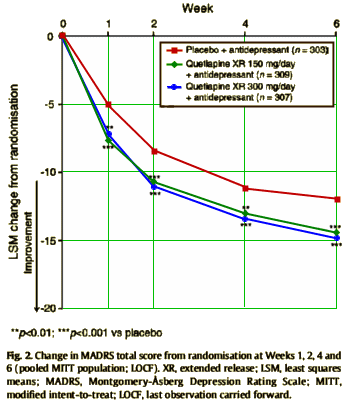
Dopamine is a feel good chemical.
Why would blocking its reception help with depression?
Duane
Take psychiatric drug studies.
Toss out the ones that are void of common sense.
And there’s not much left.
Duane
Sedation is often a much wanted, if not needed, effect. Why do so many of these drugs stop working?
“Sedation is often a much wanted, if not needed, effect. Why do so many of these drugs stop working?”
Wiley,
Because there are feedback mechanisms that regulate neurotransmitters in the synaptic cleft.
Each neuron has 19 neurotransmitters, and 19 synapses per single neuron to neuron connection. Each synapse does one neurotransmitter.
The purpose of having 19 synapses is redundancy. Some are inhibitory, some are excitatory.
The neurotransmitters complete an action that triggers or inhibits firing of the next neuron in a circuit. Neurons must fire at a proper frequency to perform something called ‘information processing’ properly.
All systems that perform information processing do it by the same basic principle. Either a transistor is on or it’s off, either a neuron fires or it doesn’t.
The synaptic clefts, chemical synapses, are self regulating closed systems with feedback. Drugs act on them as an external force. There are no mechanisms to keep the internal adjustments of one synaptic cleft uniform with another.
Take a guess what happens when time goes by. They go out of sync, and frequency of firing from one neuron in a circuit is no longer quite uniform with the next. Where the neurons are wired together to perform a particular logic (like any hard wired device) they will no longer function properly and errors occur due to timing differences where synchronization is needed.
(see: http://www.mind.ilstu.edu/curriculum/neurons_intro/neurons_intro.php for illustrations.)
The human brain is very big and made up of billions of neurons, so some can afford to malfunction without killing the patient. However, you can see the disruption on QEEG (functional tests) change with time, and acutely in response to any particular psychotropic drug.
Regulation is done by 5 mechanisms:
* adding or deleting (uncoupling) receptors,
* increasing or decreasing enzyme breakdown in the synaptic cleft
* increasing or decreasing reuptake
* increasing or decreasing release
* increasing or decreasing manufacture of the the neurotransmitter
There are other larger and and more complicated feedback mechanisms, like deleting support cells and neurons that aren’t working properly or adding more where needed. Structural changes in the brain (5-7% volume reduction over 1-2 years avg.) are seen in the brain of patients taking anti-psychotic drugs. It seems to be apoptosis of support cells (glial cells), which grow back when the drug is stopped (albeit slowly).
(see: http://www.nature.com/news/2011/110207/full/news.2011.75.html for reference)
Hope that explains that, sorry it’s so long winded.
To be honest, the state of the function of the nervous system has historically been ignored completely by American psychiatry. It’s was neither relevant to illness or treatment in the past, and still plays no role. I’ve heard neurologists say things like “those drugs screw people up”, and until recently i never asked in what way, apparently he didn’t mean subjectively.
Er, i forgot to add,
Feedback mechanisms will always overcome the effect of the drug, because of the principle of what they’re physically doing, they won’t merely meet it..
Basically that means the patient will experience symptoms of malfunction similar to withdrawal symptoms every time this (feedback overcoming the drug) occurs. Temporarily raising the dose will usually relive the subjective symptoms of this.
Drug dependence on the other hand, would be where over time the feedback becomes non-uniform throughout the brain, as receptor densities become randomly different, raising the dose would have less of an effect but may still seem to work. there are also paradoxical reactions.
At this point.. the patient is generally in for rough times ahead, particularly if their prescribing physician does not understand whats happening. This is unfortunately usually the case.
Thank you, Speck.
Yeah Speck, thank you.
Robert Whitaker goes into some detail about how D-2 receptors over-compensate when neuroleptics (dopamine antagonists) such as Seroquel are used long-term, in his book, ‘Anatomy of an Epidemic‘.
The bottom line with any of these drugs, particularly neuroleptics (aka, ‘anti-psychotics’) is that they are prescribed using subjective guess work, and their long-term use causes more harm than good, as psychiatrists like Peter Breggin and David Healey continue to point out.
Best,
Duane