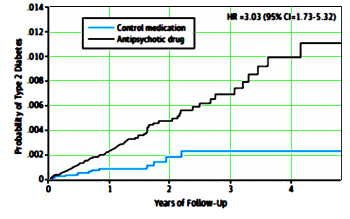
… What does it take to get clinical psychiatry to pay attention to risks that translate into actual patient harm? I fear most psychiatrists looking at the link between antipsychotics and diabetes in youth [and everyone else] will dismiss its importance. After all, diabetes is treatable. You could just throw in some metformin with an atypical antipsychotic, problem solved. And the overall health of the individual — well, that’s for the GP to deal with, not a psychiatrist.
“You could just throw in some metformin with an atypical antipsychotic, problem solved.” Unfortunately [and unsurprisingly] as this VERY concerning JAMA Psychiatry article comes out, so does the framework of an industry/academic NIMH funded paper pushing just this approach: Improving metabolic parameters of antipsychotic child treatment [IMPACT] study: rationale, design, and methods. Child and Adolescent Psychiatry and Mental Health 2013, 7:31. see http://www.capmh.com/content/7/1/31/abstract and full paper PDF here http://www.capmh.com/content/pdf/1753-2000-7-31.pdf Disclosures equally unsurprising –
Disclosures
The aripiprazole for this study was donated by Bristol-Myers Squibb.
Dr. Sikich has received funding from Bristol-Meyers Squibb for a sponsored clinical trial and donation of medication and patient travel expenses for an investigator initiated study of aripiprazole.
Dr. Correll has been a consultant, advisor, lecturer and/or data safety monitor to or has received honoraria from: Actelion, Alexza; AstraZeneca, Biotis, Boehringer-Ingelheim, Bristol-Myers Squibb, Cephalon, Desitin, Eli Lilly, Genentech, Gerson Lehrman Group, GSK, Intracellular Therapies, Lundbeck, Medavante, Medicure, Medscape, Merck, National Institute of Mental Health, Novartis, Ortho-McNeill/Janssen/J&J, Otsuka, Pfizer, ProPhase, Roche, Schering-Plough, Sepracor/Sunovion, Supernus, Takeda, Teva and Vanda. He has received grant support from BMS, Feinstein Institute for Medical Research, Janssen/J&J, National Institute of Mental Health [NIMH], National Alliance for Research in Schizophrenia and Depression [NARSAD], and Otsuka.
Dr. Hamer has been a consultant, advisor, or data safety monitor to or has received honoraria from: Abbott, Acadia, Allergan, Alkermex, Alpharma, AstraZeneca, Cenerex, Corcept, Eli Lilly, Endo, Epix, J&J, NeuroPharmaBoost, Novartis, PureTech ventures, Pfizer, Roche, Sanofi-aventis, Schwartz, Solvey, Takeda, Wyeth, and NeurogensX, Inc. Dr. Hamer and/or his spouse own stock in Bristol-Myers Squibb, Amgen, Lilly, genetech, Proctor and Gamble, and Sepracor.
Acknowledgement
The IMPACT study is funded by NIMH R01 MH080270-01A2.
There can be little question that the incidence of behavioral problems among the children on Medicaid is quite high. In fact, it would be surprising if that weren’t the case. Many of them are foster children or otherwise born into families in difficult circumstances. And for all its glitches, our foster-care system beats the orphanages of a former time hands down. These medications can be somewhat effective in situations when these children inevitably present with behavior problems, but at a very high price. Foremost, this is not a situation best approached by symptom control. Childhood is not something to be gotten through. It’s the period of complex development that shapes directions for a lifetime. This is a self-evident truth. It’s impossible to imagine that the important work of childhood can proceed through the fog of Antipsychotics – so the side effects such as the metabolic syndromes are an additional burden to children already swimming up-stream. Rampant medication in these situations is a clearly inappropriate and dangerous medical solution to a psycho-social problem. This is not simply something to to decry. It needs to be stopped.
"Aripiprazole was developed by Otsuka in Japan, and in the United States, Otsuka America markets it jointly with Bristol-Myers Squibb."
"Otsuka’s US patent on aripiprazole expires on October 20, 2014; however, due to a pediatric extension, a generic will not become available until at least April 20, 2015. Barr Laboratories [now Teva Pharmaceuticals] initiated a patent challenge under the Hatch-Waxman Act in March 2007. On November 15, 2010, this challenge was rejected by a United States district court in New Jersey."
Dr. Mickey and Dr. Rob, thank you for saying more about this.
It doesn’t matter that journal articles are based on a restrictive diagnosis. Word gets around among clinicians that metformin cancels adverse effects of atypicals, the drugs are prescribed together for whatever reason, and the world keeps turning on its axis.
Mostly, diagnosis in psychiatry is merely a justification for reimbursement. The objective is to medicate and CYA no matter what.
So many people are being arbitrarily drugged, we’re moving into a dystopian world — and lurching toward a public health disaster — that makes me very uneasy.
If only the NIMH would fund a study on Brain Volume changes in children with ongoing antipsychotic treatment. I’m sure that would get the approvals for children yanked.
Metabloic disurbances and weight gain just don’t have the same umph as saying “patients lose on average 1% of their brain volume each year. That’s an aweful lot over 18 years”
I don’t think too many people can argue brain volume reduction in children is ‘age related’ or won’t cause severe cognitive deficits to lose 1/5 of their entire brain.
The AHRP had an article titled “Did FDA Include Evidence of Brain Damage Caused by Antipsychotics”
http://www.ahrp.org/cms/content/view/606/9/ ”
The article included a new york times article “A Conversation With Nancy C. Andreasen” who’s study found the reimaining 300 of 500 patients still being followed after 18 years had brain shrinkage on an average of 1% per year. Worth noting, brain shrinkage correlated with dose, and the evidence was also supported by studys with the drugs in primates.
The evidence for brain shrinkage is just overwhelming…
In fact, Antipsychotic induced Brain Volume changes are highly reproducable, predictable by dose, and appear to correlate with severity of ilness,
http://www.ncbi.nlm.nih.gov/pubmed/20701823
http://www.ncbi.nlm.nih.gov/pubmed/22910680
http://www.ncbi.nlm.nih.gov/pubmed/22177349
http://www.ncbi.nlm.nih.gov/pubmed/21721911
http://www.ncbi.nlm.nih.gov/pubmed/9477928
Brain volume even appears to increase (reverse) when antipsychotics are stopped:
http://www.ncbi.nlm.nih.gov/pubmed/21346618
(also reproduced in animal experiments, for example
http://www.ncbi.nlm.nih.gov/pubmed/22244831 )
Subtle brain volume chnages can be detected with MRI within 6 weeks, which is within the FDA approved usage, however long term use is off-label.
I posted a comment earlier, but I hit the stop button on the browser and I have no idea if it went though (i thought it did, oh well). I had 7 replications of brain shrinkage predicatble by dose, but it’s almost off topic anyway.
NPR had a discussion about the FDA dabating the saftery and approval of antipyshciatics in children:
http://www.npr.org/templates/story/story.php?storyId=105133174
“Instead, nearly 30 percent were prescribed to kids diagnosed with attention deficit hyperactivity disorder; nearly 15 percent went to children and adolescents with no psychiatric diagnosis at all. ”
I don’t think it’s a suprise to anyone here why exactly the drug prescriptions are so high. There’s intense lobbying to brainwash and deceive just about everyone involved in approval and prescribing.
The illegal marketing and fraud lawsuits settle for millions or billions every year for almost every pharma company (a dozen last year alone!). Mostly over antipsychotics. I think our goverment already know what the problem is.. it’s that no judge is willing to give a pharama company a death sentence and convict them of the felony in such a way they can’t get goverment program reimbursement for the sale of their drugs. As it stands they just get a slap on the wrist and ‘feolony conviction’ just translates to “but here’s a ‘fig leaf’, it allows you to continue operating as you have been anyway – just pay the fine that smaller then your profits”.
I’m worried we’ll run out of children before we run out of new antipsychotics.
antipsychotics in children:*
Sorry, rubber hands today.
Off topic (I haven’t even read this post yet), but I read in a newspaper today that a former president of the American Academy of Child and Adolescent Psychiatry— William Ayres (age 81)— was just sentenced to eight years for molesting his patients over a span of three decades. I read it in The Oregonian
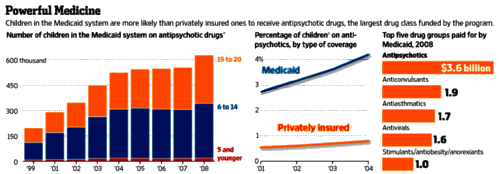
There’s money to be made even on poor people, if you can squeeze them hard enough. These drugs are 3 of the top 10 drugs prescribed on Medicaid in Illinois (and a lot of other states I bet). And many of these kids are followed by AstraZeneca right into the prison system, where Seroquel is more & more pushed to make prisoners “manageable.” It’s criminal.
Kaitlyn Bell Barnett’s book Dosed featured one of the few first person accounts I’ve seen from a young man who was doped up on Seroquel as a child in foster care. He seems to have escaped without permanent harm (?) But he recalls being too tired for school … and as a young boy just starting to try & impress the girls, humiliated and depressed to find himself growing morbidly obese.
Wiley,
I wasn’t aware they finally convicted him. He’s been hiding behind a claim of dementia for some time. Apparently, that stopped working. Like with the clergy, these psychiatrist/therapist molestation cases are always upsetting, and cast a cloud over all of us.
An older doctor who came back for training was in my residency class, and something of a friend. Some years later, he was exposed as a pedophile, longstanding, dating from his years as a GP. I went back over those residency years a jillion times trying to figure out why I didn’t know with no resolution. I even called him in prison to ask if he’d help me understand, but it was like talking to someone I didn’t really know.
I later read some things he had written in some pedophile magazine [I think called NAMBLA]. It was just a bunch of crazy rationalizations about man/boy love that made no sense. I find this very disquieting stuff that defies understanding. They design their lives and careers around being secret predators, leaving a trail of people who feel like I did – perplexed and betrayed…
It gets worse IMO…I say it’s quackery…involuntarytransformation.blogspot.com/2013/08/i-say-its-quackery.html#.Uh2puhtwoyc
It may be important to remember that the early child abuse laws were based on the NYC animal abuse statutes. Today California regulates the amount of room a chicken has in a cage while children are left to deal with life changing medications tHey do not understand and may not need.
Steve Lucas
My condolences for your situation, Mickey.
As Dr. Mickey said, “The spice must flow!” Doctors drugging children are only the last mile of the delivery system.
And this surprise from Thomas Insel:
Antipsychotics: Taking the Long View
http://www.nimh.nih.gov/about/director/2013/antipsychotics-taking-the-long-view.shtml