At first, it seems an odd candidate as a symbol for problems with the current clinical trial culture. It’s a trial at an academic center rather than one run at some commercial clinical trial center. The Principle Investigator is the Chairman of Psychiatry at Columbia, the P.I. of the widely quoted NIMH C.A.T.I.E. trial, and the immediate past president of the APA. The C.A.F.E. Trial that Dan Markingson participated in was, in fact, modeled on C.A.T.I.E. – the differences being that it was industry financed and that it focused on acute psychosis rather than chronic cases. Even more unusual for the cause of much other protest, Dan Markingson may well have been under- rather than over-medicated. But there were a few very unusual features that make it stand out:
-
Suicidal patients were excluded from the C.A.F.E. study. Dan was admitted with expressed homicidality. In psychiatry, there is no distinction between suicidality or homicidality that I’ve ever heard. Commitment laws invariably say "dangerous to self or others" in one breath. The lectures have titles like "The Lethal Patient." The claim that he was eligible because he was only homicidal is clinically absurd.
-
Dan was declared incompetent and involuntarily committed, but within days allowed to enter a voluntary drug study in lieu of going to the State Hospital. Another absurdity.
-
The outcome parameter for C.A.F.E. was voluntarily continuing the medication, yet Dan’s conditions for avoiding institutionalization were that he stay on the medication. That invalidates any reason for him to be in the study – need I say absurd once again?
-
In the treatment center where he was staying, the staff saw little to suggest any improvement. Dan’s mother increasingly worried about Dan’s clinical state and yet was told he was doing fine. Then at six months, his involuntary commitment was extended for another six months, the duration of the clinical trial. Further absurdity.
Even the most radical of antipsychiaty activists would’ve likely agreed that Dan should be tried on another drug regimen to control his ongoing and dangerous delusional state. On the face of things, it’s hard to come up with anything that would explain any of the four absurdities listed above. And the most absurd thing of all – up until the day he killed himself, he would’ve been tallied as a treatment success because he was still taking his medications.
This case has lingered for a decade, becoming a cause célèbre and it’s not because the facts of the case are unclear. It’s because the doctor involved, the chairman of his department, the medical school, and the university itself have resisted every attempt to have an independent investigation of this specific case – in spite of the efforts of Dan’s family, the bioethics department at the university, the university senate, and the scientific community at large. The university senate’s vote to investigate was turned into an investigation of the clinical trial conditions now rather than when Dan was still alive in spite of their clearly stated wishes. Carl Elliot has amassed an impressive dossier on this case [Fear and Loathing in Bioethics]. But beyond that, there’s plenty of other evidence on his site that the Markingson’s case isn’t an exception, but simply the loudest example from a program that shouldn’t be allowed to continue operating as it stands.
We’re so used to things that it doesn’t ever occur to us that there’s something very large missing in this story. It’s at the end of a long chain of misadventures. Clinical Trials are meant to be a method to as accurately as possible identify the effect of a drug under optimal controlled circumstances – in this case comparing the relative efficacy and safety of several treatments. It is hardly possible that a Clinical Trial that would allow this level of absurdity would be able to do that successfully. To my knowledge, the Principle Investigator [
Dr. Jeffrey Lieberman], the sponsoring drug manufacturer [
AstraZeneca], and the administrative contractor [
Quintiles] have remained silent. If this case isn’t a huge red flag that this clinical trial was being conducted unscientifically, I can’t imagine what it would take to make that point. Add in the evidence that the
paperwork was faked, and we have a failed study no matter how you look at things. Yet the people in charge of asking the scientific question said nothing. In fact, this study is published in the
American Journal of Psychiatry, containing the results from this site. It was meant to compare the effects of olanzapine, quetiapine, and risperidone in acute schizophrenia, and it’s not possible that the results of this study can be said to do that. Yet they concluded:
Conclusions: Olanzapine, quetiapine, and risperidone demonstrated comparable effectiveness in early-psychosis patients, as indicated by similar rates of all-cause treatment discontinuation.
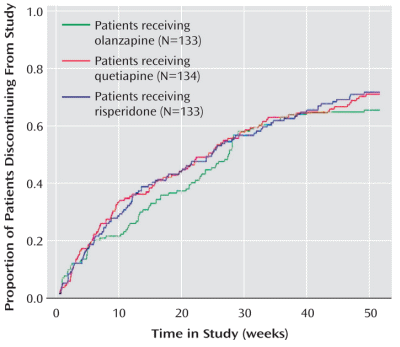
Obtaining that statement and this graph for the AstraZeneca marketing department to counter the other studies showing up Seroquel® [quetiapine] as a weak sister was the goal of the C.A.F.E. study. How can I say that without even being tentative? It’s easy, AstrZeneca said it themselves. Back in 1997, Andrew Goudie, a researcher who had previously worked on Seroquel® wrote Jeffrey Goldstein of the AstraZeneca’s R&D section requesting funding for a further study. In the return
email, Goldstein made their policies very clear:
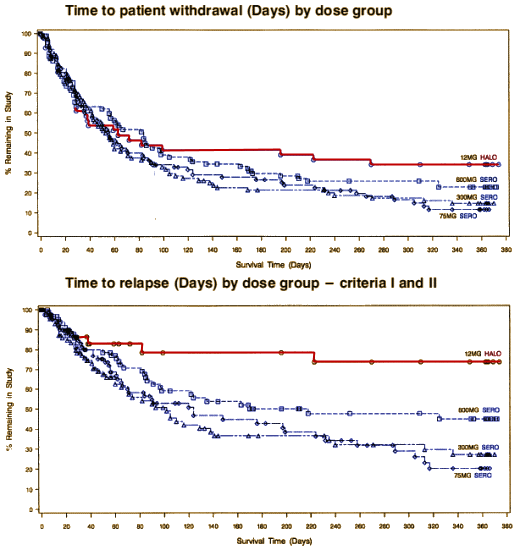
And then there was the trial known as
Study 15, very similar to C.A.F.E., except with Seroquel® [blue] versus Haldol® [red]:
In that case, they just didn’t publish the study:
Further, the flagship approval study for Seroquel® [
Trial 006] was authored by
Dr. Richard Borison, Chairman of Psychiatry at the Medical College of Georgia, who turned out to be a con man extraordinaire and ended up with a ten year prison sentence [
repressed memories…].
So Bioethicist Carl Elliot and his colleagues are completely justified in insisting that the case of Dan Markingson be investigated for gross research misconduct and unethical handling of a research subject, resulting in a death. But there’s so much more. It’s bigger than the case itself, or even the dysfunctional center where it happened. It’s a concrete symbol for an unholy alliance between a segment of academic psychiatry and the pharmaceutical industry that has made a mockery of the entire scientific enterprise we call Medicine.
 Obtaining that statement and this graph for the AstraZeneca marketing department to counter the other studies showing up Seroquel® [quetiapine] as a weak sister was the goal of the C.A.F.E. study. How can I say that without even being tentative? It’s easy, AstrZeneca said it themselves. Back in 1997, Andrew Goudie, a researcher who had previously worked on Seroquel® wrote Jeffrey Goldstein of the AstraZeneca’s R&D section requesting funding for a further study. In the return email, Goldstein made their policies very clear:
Obtaining that statement and this graph for the AstraZeneca marketing department to counter the other studies showing up Seroquel® [quetiapine] as a weak sister was the goal of the C.A.F.E. study. How can I say that without even being tentative? It’s easy, AstrZeneca said it themselves. Back in 1997, Andrew Goudie, a researcher who had previously worked on Seroquel® wrote Jeffrey Goldstein of the AstraZeneca’s R&D section requesting funding for a further study. In the return email, Goldstein made their policies very clear:

Great article.
This should be the lead story in major newspapers.
And this is the visionary who is asking us to take a “leap of faith”
First the suicide of a patient, next the suicide of a profession…
really dr mickey, thanks so much for taking the time to dig through your achives and also make these plots.
i feel they’re very informative, and i feel the type of attention the field is getting (arguably we haven’t gotten this sort of attention in 40-50 years), i think they’ll do some damage.
lets hope.
btw: really liked your earlier article talking about kanamycin and how different drugs/approaches can be used to treat similar illnesses. to me that’s what medicine is all about: there are many different ways to do something “right”, but a healthy amount of experience is often necessary before one can learn safe, effective, “alternative” solution.
Great article, right to the point. The propaganda and advertisements circulating around the University of Minnesota’s campus before and during the CAFE’ study touted the study as being a “real world” study. Supposedly, subjects could be using street drugs, drinking, whatever, it didn’t matter claimed the U’s principal investigator. An important point in Dan’s case is always overlooked, and that is the fact that he supposedly entered the study of his own free will, and volunteered his his signature on the consent form. However, when he was discharged from the hospital to a group home taking his free-will medication was not an option. The group home controlled the medication and when he took it. He was not free to discontinue from the study at any point, including the obvious court order stating so. So the point being made in the article regarding Dan being a treatment success is 100% accurate. When the outcome of a drug study is based on just exactly how long a subject can tolerate or stay on the drug, and that same subject isn’t allowed to keep his own study drug and determine if he chooses to swallow it or not on any given day is a pure mockery of data and so-called science. There just isn’t enough space to detail all of bogus and unethical or even criminal aspects of what transpired in the CAFE’ study at the U-MN.
I don’t understand how treatment success could be based on discontinuation rates. Wouldn’t reduction of symptoms be the criterion?
I could imagine that, on some level, involuntary commitment might have added to Dan Markingson’s sense of desperation, as well as having complaints going unheard. He may have felt very vulnerable, threatened, and unprotected by the essential medical neglect.
I like the use of “smoke and mirrors” in that memo from AstraZeneca. That’s a term you wouldn’t expect in connection with a “gold standard” clinical trial. I guess like sausages, the making of a clinical trial requires a strong stomach.
Alto,
You’re picking up on an important point – actually Bob Whitaker’s point. As recently as that trial [2004], the party line was that antipsychotics were for life – staving off psychosis. So the quest was for less adverse effect so people would stay on medications. Medication compliance was the gold standard. I don’t know that there’s any new consensus at this point, just opposition to meds-for-life…
I don’t get it. You mean any antipsychotic was considered to be effective as long as the person kept on taking it? That flies in the face of common sense as well as garden-variety clinical observation — and completely disregards adverse reactions that conceivably make the person worse.
Alto-
Mickey is correct. These studies were designed as a way to compare different antipsychotics. The reasoning is as follows: each of the drugs had already been tested against placebo for short term efficacy. The so-called relapse studies had established the notion that long term maintenance is optimal (since the relapse rate is higher when the drugs are stopped). So these comparison studies are assessing how long it takes before a person stops. Secondary measures include rating scales of symptoms and side effects so there is a way to try to understand why people stop but they are looking for which drugs are overall more acceptable and this is measured as time to discontinuation.
Thank you for that explanation, Sandy.
Since Dan Markingson was not permitted to stop the drug, doesn’t that make his data invalid for the study, given its objectives?
Alto,
Exactly!
Another important lesson we can learn from this tragedy is that no amount of statistical procedures, trial protocols, or ethical regulations can compensate for a lack of integrity in both researchers and clinicians. If researchers want to bend the rules to your advantage, there will always be a way for them to do so, especially if the financial benefits are measured in the millions. And where were the clinicians whose job it was to look out for Dan, to listen to him, monitor his well being, and make sure he was doing okay. His death is the result of a lack of integrity both in the system itself and in the individuals that work in that system.
Or, it makes the medication 100% effective.
OT: I once mentioned gamification as being the newest buzz in business. It seemed very out of place in a comment here. Yet, some company has already connected the dots and we have this When Doctors Play This Game, You Get Better Medical Care.
So torn. I love gaming, and I love learning. However, in the context of poor science, group think, medication escalation, and coercion, this new game unsettles me.
Wouldn’t invalidation of data associated with one of 134 quetiapine-treated subjects cause the study to be withdrawn and results recalculated?
http://www.medscape.com/viewarticle/515435
I still find it incredible that Dan Markingson was classified as responding well to treatment simply because he was still taking the drug. There’s something conceptually wrong here.