The Latin phrase deus ex machina [from deus, meaning "a god", ex, meaning "from", and machina, meaning "a device, a scaffolding, an artifice"] was referred to by Horace in his Ars Poetica, where he instructs poets that they should never resort to a "god from the machine" to resolve their plots "unless a difficulty worthy a god’s unraveling should happen"… Aristotle criticized the device in his Poetics, where he argued that the resolution of a plot must arise internally, following from previous action of the play.
The ancient advice to poets to make the resolution of their tales follow from the plot as presented to the audience remains a critical principle in literature. We want Poirot to find the murderer[s] among passenger[s] on the Orient Express, not some new outside character. We don’t like our stories ended with divine intervention or the unexpected landing of aliens. If our settlers are saved by the arrival of the cavalry, we at least want to have known they were on the way. And, by the way, that’s our beef with Clinical Trials of medications. We’d like for the reports on their efficacy and side effects to flow logically from the results of the trial itself. And that hasn’t been the case in many of the Clinical Trial reports in recent years.
History: But we didn’t know it, at least I didn’t. The Clinical Trials in question weren’t complete lies. They were subtly manipulated by the initial design, by the choice of analytic methods, by omission of data, by non-publication of negative results – a compendium of techniques that mirrored Johnny Mercer’s old lyrics:
Eliminate the negative
And latch on to the affirmative
Don’t mess with Mister In-Between
Flash forward: As the corruption of the Clinical Trial literature became more widely known, opposition arose from a variety of foci. The Clinical Trials themselves weren’t the central Achilles heel in the system, it was in the handling of the raw data – the space between unblinding the study and the publication [or burying] of the results. That was the vulnerable spot, because the pharmaceutical companies had control of access to the raw data as intellectual property [the why of that and how it came to be is beyond me]. So the consensus was that the solution to the problem was to insist that the raw data from all Clinical Trials be made available for independent analysis. Thus began the many campaigns…
An Aside: 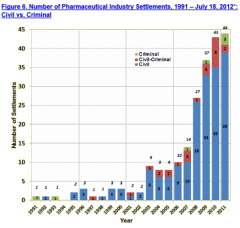 As the corruption in Clinical Trials became more widely known, the pharmaceutical companies began to take a small beating in the courts, settling more and more suits, civil [blue] and criminal [red]. But the charges were things like false advertising or off-label promotion rather than directly for deceitful data analysis. And they went out of their way to avoid retracting the targeted articles or admitting their data manipulations. They went to extremes to deny such wrong-doings, in one case even when they had essentially admitted to it in a $3 B settlement agreement [the only enduring contract…].
As the corruption in Clinical Trials became more widely known, the pharmaceutical companies began to take a small beating in the courts, settling more and more suits, civil [blue] and criminal [red]. But the charges were things like false advertising or off-label promotion rather than directly for deceitful data analysis. And they went out of their way to avoid retracting the targeted articles or admitting their data manipulations. They went to extremes to deny such wrong-doings, in one case even when they had essentially admitted to it in a $3 B settlement agreement [the only enduring contract…].
Until Last Week: As the various campaigns for Data Transparency and grown, the call has reached into governments and regulatory agencies. The pharmaceutical industry has had to make concessions, fighting the whole time to maintain their hold on the ownership of Clinical Trial raw data. They’ve argued that the reasons are patient confidentiality, protecting trade secrets, spawning innovation, and they speak as if this is some kind of fundamental right – a copyright or a property. A major breakthrough came when the European Medicines Agency [equivalent to our FDA] announced that they were going to begin releasing the Clinical Trial data once a drug was approved, and actually began doing it. It was slowed down by the suits from AbbVie and Intermune, but they were overturned and withdrawn. The European Parliment supported Clinical Trial registration in April and things were again on track for the EMA transparency initiative to move forward.
Now: Then last week, the EMA released an altogether different and very restrictive policy claiming it was little changed [to be continued…]. But that’s simply not true. It was radically changed [the U-Turn…, the end game…, a decision to reconsider…, a crushing setback…]. Did the EMA make this compromise with AbbVie to get their suit dropped? Does it have something to do with AllTrials sidling up to GSK? Did PHARMA threaten the EMA with more suits? Was this in the cards all along? Conspiracy theories abound. None of us know the answers to any of those questions. But independent analysis of Clinical Trial data sure got a lot harder in this last week – perhaps too hard. But this time, we know some things for certain. The Deus Ex Machina in this story is the pharmaceutical industry. They’re going to fight to the finish to protect their right to distort the results of Clinical Trials. And however the U Turn went down, they did it behind closed doors. It’s being presented so far by the EMA as a fait accompli, and it may well be – something of an 11th hour coup.
There’s something terribly wrong with this story. There’s a large cadre of people – doctors, scientists, healthcare advocates, and patients who are only united by an outrage at the disreputable behavior of the pharmaceutical industry in regularly distorting the scientific information from the Clinical Trials of their drugs. We want to see the raw results and are willing to put in the time – mostly unfunded – to check their analyses for accuracy. Their misbehavior has been blatant enough and damaging enough to require that kind of vigilance. And yet the European Medicines Agency is rolling out a program that makes doing that extremely difficult, has caveats that allow these companies ways to continue to hide and distort data, and have a listing of potential penalties we might endure and oaths we have to swear that are sure to discourage any but the most hearty.
Pharma’s fear is that people like me will look at their data and start to ask questions.
I am not a doctor, but when I see a sample size of 68 that is extrapolated to the general population, I have to ask questions.
When every life event or symptom, as mentioned on this blog, requires a medication, I have to ask questions.
The list goes on.
I was educated BC, before computers, We were taught that there needs to be a certain logic to any answer, not something buried so deep in some arcane mathematical calculation to make the real world application pointless, and much of what pharma has brought to market is pointless.
The real fear is that if the data is revealed many will realize the Emperor has no clothes.
Pharma has become a sham hiding behind marketing and PR and repeating this one phrase: You chose to buy the product. This logical fallacy negates any responsibility they have for the deception, and often fraud, they practice in their business model.
Steve Lucas
Thanks, Steve. Your comment and Dr. Nardo’s post serve as a gentle reminder that I really am not fine with cheating and fraud because a company thinks that is how to compete in the market. When I worked in Sales Support, I reined in salespeople more than once. Gratefully, I never saw outright fraud or even intent in our sales people. Most of the time it was from cutting corners or because they wanted to advertise what our competitors were, and they couldn’t understand why I wasn’t about to put what they requested on our websites. I worked with Compliance on almost a daily basis and our divisions lawyers on every project. I once worked with one of the corporation’s lawyer when the AG was investigating my product, industry-wide. It was educational, but I can’t say that I enjoyed that experience.
As far as not buying what a company is selling, I don’t believe in boycotts, but where I spend my money is the only vote I have, so I’ll keep trying to make the most informed decisions I can. I’m far from a radical, and not even an activist, yet I am no different than in my early 20’s when my friends wanted me to smoke weed with them and I said no. When I told them one of the reasons was that they had no idea where their pot came from, maybe somebody died smuggling it in, they assured me it was homegrown. No, it wasn’t, and they paid good money for it not to be. Sorry to digress. This was only to illustrate that I have always considered the hidden costs of things, and although I’ve learned that I can’t do a damn bit about most of them, I can choose not to spend money on some of them.
Great post Mickey. I do hope we are both young enough to be able to wait to find out the solution to this plot.
Some of the data may be suspect, yet the platform holds hope
FDA launches openFDA.
openFDA!! The FDA doesn’t do anything with the adverse events reports it’s been collecting for decades. It doesn’t have the data-mining tools. “Post-marketing surveillance” is nothing but an empty phrase. Nobody’s surveilling, the FDA’s database might as well be a giant digital trashcan.
Knowing this, nobody takes making FDA adverse event reports seriously.
So, a few third parties, such as Rxisk.org and AdverseEvents.com, got FDA APIs some years ago and started playing around with the data. (Rxisk.org is David Healy’s pro bono project; AdverseEvents.com sells reports to industry.)
Now the FDA is making public relations hay out of a non-event, openFDA. Oh, some apps based on the AER database? So freakin’ what. What would you expect to get out of it? The database is spotty, the reports are anecdotal, the FDA couldn’t care less. Maybe somebody else can do something with this stuff US taxpayers have paid $$ millions to collect, and the FDA can point to this high-tech accomplishment.
Ok, call me naive; I am hoping it will generate interest in the subject. Of course data is key. But that is not what I see hopeful about it at the present moment. I would very much like the technical crowd to take an interest in this subject. Not for another consumer app, although that would be handy for me, but to grow the demand for access to information.